A systematic review of the safety and efficacy on cognitive function of herbal and nutritional medicines in older adults with and without subjective cognitive impairment.
Study Design
- Type d'étude
- Systematic Review
- Population
- Older adults
- Durée
- 12 weeks
- Intervention
- A systematic review of the safety and efficacy on cognitive function of herbal and nutritional medicines in older adults with and without subjective cognitive impairment. None
- Comparateur
- placebo
- Critère de jugement principal
- cognitive function
- Direction de l'effet
- Positive
- Risque de biais
- Low
Abstract
BACKGROUND: Subjective cognitive impairment (SCI) substantially increases dementia risk and is often conceptualised as the preclinical asymptomatic phase of the cognitive decline continuum. Due to the lack of pharmacological interventions available to treat SCI and reduce dementia risk, and the popularity of herbal and nutritional medicines, the primary aim of this review was to investigate the efficacy on cognitive function and safety of herbal and nutritional medicines (relative to a control) for older adults with and without SCI. The secondary aims were to describe the study characteristics and assess the methodological quality of included studies. METHOD: Five databases (Cochrane, MEDLINE, CINAHL, PsycInfo, and EMBASE) were searched from database inception with weekly alerts established until review finalisation on 18 September 2022. Articles were eligible if they included the following: study population of older adults with and without SCI, herbal and nutritional medicines as an intervention, evaluated cognitive outcomes and were randomised control trials. RESULTS: Data were extracted from 21/7666 eligible full-text articles, and the risk of methodological bias was assessed (with SCI = 9/21; without SCI = 12/21). Most studies (20/21) employed parallel, randomised, placebo-controlled designs and were 12 weeks in length. Herbal supplements were widely used (17/21), namely a form of Ginkgo biloba (8/21) or Bacopa monnieri (6/21). Measures of cognition varied across studies, with 14/21 reporting improvements in at least one domain of cognitive functioning over time, in the intervention group (compared to control). A total of 14/21 studies were deemed as having an overall high methodological risk of bias, 6/21 had some concerns, and only one study (using an SCI population) was assessed as having a low risk of methodological bias. CONCLUSIONS: Overall, this review found that there is a low quality of evidence regarding the efficacy of cognitive function and safety of herbal and nutritional medicines for older adults with and without SCI, due to a high risk of bias across studies. Additionally, further work needs to be done in classifying and understanding SCI and selecting appropriate trial primary outcomes before future studies can more accurately determine the efficacy of interventions for this population.
En bref
There is a low quality of evidence regarding the efficacy of cognitive function and safety of herbal and nutritional medicines for older adults with and without SCI, due to a high risk of bias across studies.
Full Text
Systematic Reviews
RESEARCH Open Access
A systematic review of the safety and efficacy on cognitive function of herbal and nutritional medicines in older adults with and without subjective cognitive impairment
Adele E. Cave1, Dennis H. Chang1, Gerald W. Münch1,2 and Genevieve Z. Steiner‑Lim1,3*
Abstract
Background Subjective cognitive impairment (SCI) substantially increases dementia risk and is often conceptual‑ ised as the preclinical asymptomatic phase of the cognitive decline continuum. Due to the lack of pharmacological interventions available to treat SCI and reduce dementia risk, and the popularity of herbal and nutritional medicines, the primary aim of this review was to investigate the efficacy on cognitive function and safety of herbal and nutri‑ tional medicines (relative to a control) for older adults with and without SCI. The secondary aims were to describe the study characteristics and assess the methodological quality of included studies. Method Five databases (Cochrane, MEDLINE, CINAHL, PsycInfo, and EMBASE) were searched from database inception with weekly alerts established until review finalisation on 18 September 2022. Articles were eligible if they included the following: study population of older adults with and without SCI, herbal and nutritional medicines as an interven‑ tion, evaluated cognitive outcomes and were randomised control trials. Results Data were extracted from 21/7666 eligible full‑text articles, and the risk of methodological bias was assessed (with SCI = 9/21; without SCI = 12/21). Most studies (20/21) employed parallel, randomised, placebo‑controlled designs and were 12 weeks in length. Herbal supplements were widely used (17/21), namely a form of Ginkgo biloba (8/21) or Bacopa monnieri (6/21). Measures of cognition varied across studies, with 14/21 reporting improvements in at least one domain of cognitive functioning over time, in the intervention group (compared to control). A total of 14/21 studies were deemed as having an overall high methodological risk of bias, 6/21 had some concerns, and only one study (using an SCI population) was assessed as having a low risk of methodological bias. Conclusions Overall, this review found that there is a low quality of evidence regarding the efficacy of cognitive function and safety of herbal and nutritional medicines for older adults with and without SCI, due to a high risk of bias across studies. Additionally, further work needs to be done in classifying and understanding SCI and selecting appropriate trial primary outcomes before future studies can more accurately determine the efficacy of interventions for this population.
*Correspondence: Genevieve Z. Steiner‑Lim [email protected] Full list of author information is available at the end of the article
© The Author(s) 2023.Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visithttp://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativeco mmons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Keywords Cognition, Complementary medicine, Herbal medicine, Nutrition, Subjective cognitive impairment (SCI), Mild cognitive impairment (MCI), Dementia, Alzheimer’s disease, Systematic review
Introduction
Subjective cognitive impairment (SCI) is a self-perceived worsening of cognitive functioning, particularly in the area of memory, that cannot be verified by neuropsychological tests [1, 2]. SCI lies on a continuum of healthy cognitive ageing and is conceptualised as the preclinical phase of dementia (healthy cognitive ageing, to preclinical SCI, followed by prodromal mild cognitive impairment (MCI), then dementia) [2–4]. SCI is estimated to double the risk of future objective decline (MCI or dementia) [5, 6], carries an increased prevalence of Alzheimer’s disease biomarkers and impacts mental health (1 in 3 people) and functional ability (1 in 2 people) [7], making it an important area of focus for secondary prevention research and care.
It is estimated that the prevalence of SCI is 1 in 4 older adults aged 60 years and above, worldwide, with these numbers increasing rapidly each year [2]. Currently, there are no approved pharmacological interventions available, with many older adults experiencing SCI seeking alternative treatments [8]. Difficulty also lies with the assessment of SCI, as current diagnostic tools have been developed for MCI or dementia [8, 9]. Furthermore, inconsistencies in the categorisation of SCI (namely the division between healthy adults without SCI and those with SCI) are apparent in research [8, 9]. Due to the increased risk of dementia and high prevalence of SCI, high-quality research into effective treatments to improve cognitive functioning and prolong further decline is needed.
A review and meta-analysis conducted in 2018 investigated a variety of interventions (group psychological, cognitive, lifestyle and complementary and alternative medicines) for the treatment of SCI and their efficacy on psychological well-being, metacognition and objective cognitive performance [9]. The authors found that studies were generally of low quality; hence, no firm conclusions could be made about the efficacy of the interventions employed [9]. Whilst this review/meta-analysis is of great importance to furthering SCI treatment research, it did not explore the efficacy of single interventions on cognitive functioning, nor did they investigate this usage and efficacy in older adults without SCI.
Complementary medicines (CMs) are defined as a broad range of health care approaches that are not thought to be part of conventional medical care [10, 11]. CMs are classified into three primary categories of delivery: nutritional (e.g. herbs, dietary supplements), psychological (e.g. meditation, relaxation therapy) and
physical (e.g. acupuncture, massage) [10]. CMs are becoming more widely available and used by older adults, particularly herbal and nutritional medicines for the treatment of chronic health conditions including, cardiovascular disease [12], diabetes [13] and dementia [14, 15]. Herbal medicines contain herbal substances or herbal preparations, with nutritional supplements/medicines containing vitamins, minerals and in combination formulas and herbal substances/preparations as well [11, 16]. The natural properties of these medicines make them attractive to individuals wanting to improve their general health and well-being [11].
The primary aim of this review was to investigate the efficacy of cognitive function and safety of herbal and nutritional medicines (compared to an appropriate control group) for older adults with and without SCI. The secondary aims were to describe the study characteristics and assess the methodological quality of included studies, utilising the Cochrane risk of methodological bias (ROB 2) tool. This is the first review, to our knowledge, that has investigated the use of herbal and nutritional medicines for older adults with and without SCI, in depth.
Methods
This review is structured according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [17] and registered with the PROSPERO international database of systematic reviews on 7 May 2021 (#CRD42021244631). A protocol was not published for this review.
Eligibility criteria
A scoping review was conducted in line with the study eligibility criteria which were determined as per the PICOS principles for systematic reviews [18]:
- 1. Population: older adults1 with and without subjective cognitive impairment (subjective cognitive impairment is a self-perceived worsening of cognitive functioning) [1, 2]
- 2. Intervention: herbal and nutritional medicines (herbal medicines containing herbal substances or herbal preparations, and/or nutritional supplements/
1 Older adults (with and without SCI) were defined as aged 45 years and older, in accordance with the US Centers for Disease Control and Prevention (CDC) population-based statistics on Subjective Cognitive Decline and Aging [7].
- Table 1 Keywords forming the search strategy of the review utilised for the five databases
Area and search number Search terms Subjective cognitive impairment (S1) “subjective cognitive impairment” OR “SCI” OR “sub‑
jective cognitive complaint*” OR “SCC” OR “subjec‑ tive memory complaint*” OR “SMC” OR “cognitive decline” OR “preclinical dementia” OR “preclinical Alzheimer*” OR “age associated cognitive decline” OR “age related cognitive decline” OR “age associ‑ ated memory impairment”
Older adults without subjective cognitive impairment (S2) “healthy ageing” OR “healthy aging” OR “older adult*” Intervention (S3) “herbal medicine” OR “Chinese medicine” OR “com‑
plementary medicine” OR “alternative medicine” OR “natural medicine” OR “vitamin* OR nutraceu‑ tical” OR “nutritional supplement” OR “Chinese herbal medicine” OR “traditional Chinese medicine” OR “ginkgo” OR “ginseng” OR “alpha‑lipoic acid” OR “lipoic acid” OR “bacopa monnier*” OR “brahmi”
(S4) S1 OR S2 AND S3
*indicates truncation
medicines containing vitamins, minerals, fatty acids etc., separately or in combination formulas) [11, 16]
- 3. Comparisons: appropriate control group (non-active orally ingested placebo, orally ingested active control)
- 4. Outcome: measures of cognition (both standardised/ validated and non-standardised/non-validated testing measures)
- 5. Study design: randomised control trials (parallel or cross-over)
The following are the inclusion criteria: chronic dosing studies over a period of 2 weeks or more, peer-reviewed articles fully accessible online and written in English that met the above PICOS criteria. The following are the exclusion criteria: reviews, case studies, editorials, conference proceedings, preclinical studies (both in vitro and in vivo), trial protocols, trial registrations, book chapters, abstracts only, peer-reviewed articles in which the study population had a diagnosis of mild cognitive impairment or dementia, did not include cognition as a primary or secondary endpoint, or employed a co-intervention such as cognitive training.
Search strategy
Two researchers (AEC, GZS) reviewed the search strategy in consultation with an experienced librarian, prior to the commencement of scoping. Four databases were searched for peer-reviewed articles: Cochrane, MEDLINE, CINAHL and PsycInfo from inception to 4 August 2018, and a further fifth database, EMBASE, was searched on 14 September 2022. Weekly alerts were established across the five databases until review finalisation on 18 September 2022. A full list of keywords is detailed below in Table 1. The only modification to the
search strategy was the exclusion of non-randomised controlled trials from the Cochrane database to reduce the number of records for screening. Reference lists of included studies were also searched to identify any further eligible studies. Studies that included multiple age groups were also included if they reported demographics and outcomes separately for older participants in line with the eligibility criteria.
Data extraction and appraisal
All titles and abstracts were first screened by one author (AEC) for inclusion or exclusion from the review. If there were uncertainties regarding suitability for inclusion, the second reviewer (GZS) would assist to collaboratively make a final decision. Full-text articles were reviewed by the two authors with disagreements of acceptability resolved by discussion. Study characteristics were then extracted for each full-text article. These characteristics included author(s) and study location, aim, study population (group, sex, mean age, standard deviation and range), diagnosis criteria/global cognition measure, study design and outcome measurement frequencies, intervention, dose and duration, measures of cognition and results (cognition, retention, adherence and adverse events).
A methodological risk of bias assessment was conducted in accordance with the Cochrane Review Process for Randomised Trials (ROB 2) [19]. The quality of trial design, conduct and reporting of the included studies was assessed. Separate risk of bias assessments was conducted for parallel [20–39] and cross-over design studies [40]. The risk of bias tool evaluates five domains: bias arising from the randomisation process, deviations from intended interventions (effect of assignment and
adherence to intervention), bias due to missing outcome data and in the measurement of the outcome, and bias in the selection of the reported result [19]. The sixth domain of bias arising from period and carryover effects was also evaluated for the cross-over study [19]. One author (AEC) independently conducted the risk of bias assessment, with the second author (GZS), reviewing the outcomes.
Individual studies were assessed as low risk, some concerns regarding methodology and high risk based on each of the above-mentioned domains. Studies with one or more domains assessed as high risk or with some concerns for multiple domains were deemed overall as high risk. Those with at least one domain with some concerns were evaluated in this category. The risk of bias process was conducted to assess the methodological quality of studies in their published form; study authors were not contacted for further information. A qualitative synthesis approach to this review was taken due to the large variation of interventions and cognitive assessments utilised across the studies, for each of the populations.
Results
Study selection
Figure 1 outlines the study selection process, with twenty-one studies meeting the eligibility criteria [20– 40]. Nine studies involved older adults with SCI [20–22,
- 26, 28, 33, 35, 37, 38], and the remaining twelve, older adults without SCI [23–25, 27, 29–32, 34, 36, 39, 40].
Fig. 1 PRISMA flow diagram illustrating the study selection process
Study characteristics
- Table 2 details a summary of the characteristics of the nine [20–22, 26, 28, 33, 35, 37, 38] SCI studies, and
- Table 3 details the twelve studies in older adults without SCI [23–25, 27, 29–32, 34, 36, 39, 40]. Both tables outline the study aim, population (group, sex, mean age, standard deviation and range), diagnosis/global cognition measure, design, intervention and dose, duration, measures of cognition and results (cognitive outcomes, retention and adherence, and adverse events).
Across both populations, all studies were randomised, double-blind, placebo-controlled trials [20–40]. Twenty studies employed a parallel design [20–39], and one study utilised a cross-over design [40]. Three studies utilised the same intervention with two different doses (three comparison groups in total, including placebo) [20, 22, 28], and one study utilised two different interventions compared to a single control [29].
Eight studies were conducted in the USA [24, 25, 27–31, 36], three each in Australia [23, 32, 38] and Italy [26, 33, 39], two in India [21, 35] and one each in Korea [20], the Netherlands [22], the UK [34], China [37] and
Japan [40]. Two studies were published from 1995 to 1998 [22, 33], ten published between 2000 and 2010 [21, 23–25, 27, 30–32, 35, 36], with the remaining nine between 2012 and 2020 [20, 26, 28, 29, 34, 37–40].
Fifteen studies reported methods of recruitment [20, 22–26, 28, 29, 32, 35–40], with twelve studies conducted in community settings (audio, visual, and print media, universities) [20, 23–25, 28, 29, 32, 35–38, 40] and three in clinical settings (general practice and outpatient clinics) [22, 26, 39].
Participants
Across the included studies, the total sample size (at baseline) was N= 1891, with 19/21 studies reporting participant sex (N= 1798; 61% were female) [20– 25, 28–40]. The mean age of participants reported across 20/21 studies was 65.43 years [20–34, 36–40], SDpooled= 13.95 (for 17 studies) [20–23, 25, 27–33, 36–40]. Individual studies ranged from 28 [40] to 262 participants [31]. A total of 755 participants were from the nine SCI studies [20–22, 26, 28, 33, 35, 37,
- Table 2Summary of characteristics for studies involving older adults with subjective cognitive impairment
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
Retention/adherence:
nificant improvement
compared to placebo
to placebo with a sig‑
completed the study:
reported and no par‑
adherence rate in all
memory and execu‑
no serious AE’s were
Cognition: improve‑
93.3%, with a > 90%
ment in short‑term
in both HD and LD
ticipants withdrew
69/75 participants
groups compared
90% and placebo
in the HD group
Adverse events:
HD 93.3%, LD
tive function
due to AE’s
groups
alone
span task (CANTAB),
executive function
8 weeksImmediate recall
tern recognition
and the spatial
from the pat‑
memory task
(WCST)
2) LD = 600 mg daily
3) Matched placebo
(species of fungus)
(2 capsules, 3 × per
(2 capsules, 3 × per
day) N.R = placebo
Tremella fuciformis
daily (2 capsules,
1) HD = 1200 mg
3×per day)
ingredients
day)
trial, outcomes at 0
placebo control
double‑blind
scale ≤ 0.5 Randomised,
and 8 weeks
MMSE (≥ 25), clinical
dementia rating
(65F:10 M) with sub‑
complaints aged
jective cognitive
in the HD group
53.83 (± 5.5)m
in the LD group
dwelling adults
in the placebo
with 26F:4 M
and 26F:4 M
and 13F:2 M
Community
years, range
40–65 years
group
tion ofTremella fuci-
[20] To examine the effi‑
in individualsformis
of oral administra‑
cognitive impair‑
cacy and safety
with subjective
ment
Ban et al. 2018Korea
Table 2(continued)
Retention/adherence:
then excluded as out‑
placebo in: digit span
Cognition: significant
tion were considered
paired associates dis‑
liers, participants tak‑
group × time interac‑
ing ≥ 85% of medica‑
or moderate adverse
similar delayed recall
backward, list learn‑
and visual retention
tion (improvement)
for interventionc.f.
ing delayed recall,
the study with 15
59/65 completed
events reported
no serious, mild
Adverse events:
by participants
compliant
cognition Results
(based on pictures),
digit symbol (WAIS)
(based on designs),
Digit Span Forward
and delayed recall,
pairs—immediate
(WMS‑1) immedi‑
Serial Subtraction
associates—simi‑
Cancellation Test,
visual retention 1
visual retention 2
lar and dissimilar
(RAVLT) immedi‑
Test, list learning
ate and delayed
ate and delayed
recall, passages
and Backward
control and dose DurationMeasures of
recall, paired
(WAIS), Digit
12‑week interven‑
tion period then
a 12‑week with‑
drawal period
only states ‘matched
with bioactive con‑
®BacoMind(stand‑
ofBacopa monnieri
stituents) 1 × 450‑
Matched placebo
mg capsule daily
1×capsule daily,
ents in placebo,
without actives’
ardised extract
measure DesignIntervention/
N.R = ingredi‑
outcomes at 0, 12
double‑blind pla‑
cebo control trial,
and 24 weeks
MMSE (≥ 24)Randomised,
study location AimStudy populationDiagnosis criteria/
global cognition
nitive deficits (AAMI)
with no major cog‑
range 50–75 years
Adults with mem‑
aged=64.98m
ory complaints
with 23F:42 M
(± 9.37) years,
for 1 year
the clinical efficacy,
®Mindon impaired
memory in elderly
safety and toler‑
ability of Baco‑
2008[21]India To evaluate
individuals
Barbhaiya et al.,
Author and
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
participants withdrew
cebo = 15 (total reten‑
Retention/adherence:
headache and sleepi‑
complaints, dizziness,
1 and 2 tests. Within‑
on the EMCT or Rey
increase in the low‑
ing but not limited
to gastro‑intestinal
with an adherence
Adverse events: 25
group, Benton test
due to AEs includ‑
dose = 14 and pla‑
ments across time
tion rate of 81.7%)
nificant between‑
Cognition: no sig‑
total withdrawals
scores increased
group improve‑
with the largest
group = 15, low
from high dose
ness (across all
rate of 93.98%
in all 3 groups
dose group
groups)
visual memory), Rey
memory and learn‑
revised (short‑term
of visual retention‑
(short‑term verbal
test parts 1 and 2
tion), Benton test
and concentra‑
treatment EMCT (measur‑
ing, long‑term
ing attention
memory)
4 weeks washout
then 24 weeks
leaf, folium, ethanol,
both solutions were
chlorophyll was dis‑
soluble chlorophyll
flavone glycosides)
drops, 1.9 ml undi‑
tion:Ginkgo biloba
powder dissolved
extract (composi‑
solved in alcohol)
1) High dose (40
3×daily) (water‑
extract with pla‑
taste and colour
(water insoluble
mixed together,
2) Low dose (40
of all 3 dosages
luted 3 × daily)
water mixture,
cebo 3 × daily)
3) Placebo (40
alcohol/water
in an alcohol/
Ginkgo biloba
drops (1.9 ml
drops, 1.9 ml
similar
outcomes at 0, 6, 12
double‑blind pla‑
cebo control trial,
and 24 weeks
of known origin Randomised,
and memory loss
MMSE (≥ 20)
group and 47F:35 M
with self‑reported
=68.96 (± 7.77)m
group, 50F:32 M
tion complaints
and 46 females
in the placebo
in a high‑dose
with 31 males
memory and/
or concentra‑
in a low‑dose
Elderly adults
years, range
55–86 years
group
of an alcohol/water
with memory and/
cebo in the elderly
versus pla‑biloba
extract ofGinkgo
or concentration
of two dosages
and tolerance
[22]lands To determine
the efficacy
complaints
1998The Nether-
Brautigam et al.,
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
with 95% adherence
ment in MMSE score
Adverse events: one
8 weeksMMSECognition: improve‑
after active product
ence: no withdraw‑
and 90% in the pla‑
in the active group
in the active group
reported aftertaste
Retention/adher‑
als from the trial,
cebo group
intake
mcg, vitamin B12 33
1×capsule per day,
dry extract 320 mg,
capsule form and 1
9.5 mg, Biotine 450
ofBacopa monnieri
mcg, copper 2 mg,
mcg, folic acid 400
mcg, vitamin D 25
Matched placebo,
30 mg, vitamin B6
‑Teanina 100 mg,
N.R = placebo
Crocus sativus
Combination
ingredients
per day
trial, outcomes at 0
placebo control
double‑blind
Randomised,
and 8 weeks
cognitive decline
diagnosis of cog‑
without a known
MMSE (20–27),
self‑perceived
nitive decline
or dementia
of age) (N.R = range,
perceived cognitive
(=66 (± 3) yearsm
30 older adults
sex), with self‑
decline
of a rational assem‑
functions in a sam‑
blage of nutraceu‑
ticals on cognitive
ple of 30 elderly
[26]Italy To evaluate
the effects
subjects
Cicero et al., 2016
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Retention/adherence:
and 600 mg interven‑
group due to adverse
Adverse events: three
participants reported
drew (1 from the pla‑
spearmint group sig‑
deemed ‘not related’
(900 mg) adherence
events with a 98.1%
in both the placebo
in working memory
and spatial working
(600 mg) and 100%
3 participants with‑
nificantly improved
myalgia, headache,
Cognition: 900 mg
and heartburn. All
memory accuracy
cebo group and 2
worsening of oily
tion of heartburn
scalp, cystic acne
from the 600 mg
(placebo), 99.1%
with the excep‑
AE’s: knee pain,
tion groups
and tracking (motor
investigating atten‑
90 daysCDR − with 11 tasks
episodic and work‑
executive function
tion and informa‑
tion processing,
ing memory,
control)
crystalline cellulose/
3) Placebo capsules
(L.)/Mentha spicata
(L.)/Mentha spicata
matched on taste,
spearmint extract
spearmint extract
contained micro‑
day (2 × capsules
day (2 × capsules
day (2 × capsules
total), N.R = if
appearance
1) 600 mg
2) 900 mg
and smell
total)
total)
(participants arrived
equiv. to the overall
on testing day fast‑
and 90 days (aver‑
ing from product/
score for that day)
across testing day
outcomes at 0, 45
Average of scores
double‑blind pla‑
cebo control trial,
age on each day)
tion of interven‑
assessed 15 min
and 2, 4 and 6 h
tion or placebo,
after ingestion.
food and were
prior to inges‑
Randomised,
then 30 min
MAC‑Q (≥ 25), WMS
(≤ 29 on VPA‑1)
and WMS (≤ 9
MMSE (≥ 24),
on VPA‑2)
50–70 years with 30
participants in each
spearmint), (overall
(± 0.6) years, range
Institute of Mental
Adults with AAMI
mint and 900 mg
group (placebo,
on the National
Health criteria)
600 mg spear‑
aged=59.4m
30 M:60F)
(based
extract on cognitive
performance, sleep
the effects of sup‑
and mood in indi‑
viduals with AAMI
with a spearmint
2018[28]USA To investigate
plementation
Herrlinger et al.,
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Retention/adherence:
pants < 80% excluded
nausea and vomiting,
ence was an average
1/56 non‑compliant)
of 97%, with partici‑
and one in placebo
cantly for the inter‑
ticipants withdrew
Adverse events = 1
from the trial (only
across time, com‑
developed a mild
working memory
Cognition: spatial
51/56 completed
the study. Adher‑
pared to placebo
tion experienced
improved signifi‑
vention group,
rash, both par‑
from interven‑
reaction time
participant
from study
ment battery (SUC‑
16 weeksSwinburne Univer‑
(word list learning)
sity computerised
cognitive assess‑
CAB) and CVLT‑2
with 46 ingredients,
seed), 1 tablet each
dry fruitmarianum
B2), 1 tablet taken
(St. Mary’s thistle),
and 2 mg of ribo‑
contained starch
tablet (combina‑
quantities being
Swisse Women’s
andVitis vinifera
dry seed (grape
with the largest
day with break‑
tablet matched
of intervention,
on appearance
flavin (vitamin
with breakfast
™Ultivite 50 +
Ginkgo biloba
fromSilybum
fast. Placebo
tion formula
trial, outcomes at 0
placebo control
and 16 weeks
double‑blind
Randomised,
with a medical prac‑
MMSE (≥ 24), mem‑
mine whether par‑
to screen for SMCs
and medical exam
ory questionnaire
titioner to deter‑
in good health
ticipants were
memory complaints
64–82 years with 28
participants in each
multivitamin group
(± 4.3) years, range
group (=70.3m
(=71.9 (± 4.8)m
Women report‑
years, placebo
ing subjective
group
tivitamin and herbal
mentation of a mul‑
tion in community‑
of 16‑week supple‑
formula on cogni‑
dwelling, elderly
2012[38]Australia To investigate
the effects
women
Macpherson et al.,
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
increased in the treat‑
compared to placebo
Adverse events: none
(reported at 3‑month
ence: no withdrawals
reported in the study
ence rate as an aver‑
age across the dura‑
Cognition: memory
reported by partici‑
with a 92% adher‑
index significantly
Retention/adher‑
tion of the study
at the endpoint
ment group
intervals)
pants
recall and deferred
index, acquisition
9 monthsRMT − (memory
memory)
to study drug—two
capsules daily (one
capsules daily (one
bitartrate, minerals
seng extract G115
Placebo ‘identical’
(N.R = dose) (from
Standardised gin‑
and vitamins (not
), dimeth‑ginseng
the root ofPanax
one after lunch).
one after lunch),
specified)—two
ylaminoethanol
after breakfast,
after breakfast,
N.R = placebo
ingredients
MMSE (≥ 24)Randomised, dou‑
3, 6 and 9 months
ble‑blind placebo
comes at 15 days,
(only first and last
control trial, out‑
session analyses
conducted)
with AAMI, 21F:9 M
group and 21F:9 M
in the intervention
(=60.45 (± 3.9)m
in the placebo
51–65 years)
Older adults
years, range
group
well‑being—affec‑
interrelationships
domains of well‑
tive functioning
being and their
tion and cogni‑
(psychological
and perceived
(intervention)
on two major
quality of life)
[33] To determine
the influence
of treatment
Neri et al., 1995Italy
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
patient withdrew due
ence: 87.5% retention
in the placebo group
ing the intervention
and 12 weeks), digit
nificantly improved
forward (12 weeks)
at each of the time
control (12 weeks),
rashes in the inter‑
and 12 weeks) sig‑
and 1 in the SBME
and paired associ‑
Cognition: mental
to maculopapular
(4 weeks, 8 weeks
Adverse events: 1
group withdrew),
Retention/adher‑
points listed dur‑
N.R = adherence
ated learning (8
logical memory
vention group
(2 participants
period
visual reproduction
ate learning (WMS)
and paired associ‑
logical memory,
digit backward,
of a placebo period Mental control,
digit forward,
following 4 weeks
12 weeks, then
Placebo twice daily,
of administration.
N.R = administra‑
125 mg of SBME
tion, ingredients
twice a day,
N.R = type
outcomes at 0, 4, 8,
double‑blind pla‑
cebo control trial,
12 and 16 weeks
aMemory (< 6) Randomised,
and WMS Logical
MMSE (≤ 24)
with 20 participants
with AAMI, range
(± SD), (3F:37 M)
N.R = mean age
in each group
55–70 years,
Forty adults
[35]India To study the effi‑
cacy of SBME
in subjects
with AAMI
Raghav et al., 2006
Table 2(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
Retention/adherence:
adverse event reports
and memory retrieval
group, both interven‑
completed the study,
group they withdrew
Cognition: no signifi‑
retrieval for placebo)
cant between group
across time—audio/
outcomes improved
98/101 participants
of participants due
across time. Within
upset, N.R = which
to gastrointestinal
abstracting ability
Adverse events: 2
tion and placebo
(except memory
with withdrawal
N.R = adherence
visual memory,
improvements
from
memory, abstract‑
ing and memory
12 weeksVisual/auditory
recall
dose measurements
,Catharanthus roseus
capsules with meals
capsules with meals
leaf), acetyl‑sinensis
placebo ingredients
p/day) or additional
‑carnitine, cola nutl
extract (),kola nitida
vitamin B6, vitamin
flour, N.R = specific
mainly containing
Matched placebo,
for active and pla‑
choline bitartrate,
and‑cysteine) (2l
Advanced (Phos‑
extract (Camellia
‑phenylalaninel
Huperzia serrata
‑pyroglutamicl
B12,‑tyrosine,l
acid, green tea
phatidylserine,
cebo (states 2
,Ginkgo biloba
(whole plant),
‑glutamine,l
BrainPower
daily
trial, outcomes at 0
placebo control
and 12 weeks
double‑blind
Randomised,
gorised as ‘severe SC
(moderate severe), 4
as ‘slight SC group’, 3
SC) or 2 (slight/mild
SC) were combined
for a Chinese popu‑
(severe) and 5 (very
or occasional slight
tion/concentration
lation, participants
group’, N.R = name
severe) were cate‑
or impaired atten‑
medical question‑
naire, constructed
of questionnaire
scoring 1 (no SC
of hypomnesis,
as determined
and validated
by a standard
forgetfulness,
memory loss
No reports
group and 34F:17 M
numbers calculated
(± 10.5) years, range
including 33F:14 M
in the intervention
unteers with SMCs
after withdrawals)
47.28–88.43 years
Community vol‑
and participant
in the placebo
aged67.1m
group (ages
the safety and effec‑
term administration
dietary supplement
a multi‑ingredient
tiveness of short‑
on SMCs in older
of BrainPower
[37]China To evaluate
Advanced,
adults
Zhu et al., 2016
AAMIAECANTABCDRCVLT-2EMCTAge-associated memory impairment,Adverse event,Cambridge Neuropsychological Test Automated Battery,Cognitive drug research system,California Verbal Learning Task,Expended
SBMEBacopa monnieriSCSMCsVPAWAISWCSTWMSStandardisedextract,Symptom complaints,Subjective memory complaints,Verbal paired associates,Wechsler Adult Intelligence Scale,Wisconsin Card Sorting Test,
HDLDMAC-QMMSEN.RRAVLTRMTMental Control Test,High dose,Low dose,Memory Complaint Questionnaire,Mini-Mental State Examination,Not reported,Rey Auditory Verbal Learning Test,Randt Memory Test,
aRaghav et al. (2006) [35] reported on AAMI without any evidence of dementia or psychiatric disorder despite excluding individuals scoring > 24 on the MMSE
Weschler Memory Scale
- Table 3Summary of characteristics for studies involving older adults without subjective cognitive impairment
Retention/adherence:
nificant improvement
adherence measured
reported by 2 partici‑
withdrew from study
pants who withdrew
13 participants with‑
storage and retrieval
in the ginkgo condi‑
and post‑treatment,
tasks for theBacopa
and digestive prob‑
ence: 6 participants
in placebo group, 9
drew between pre‑
Adverse events: 10
in the intervention
ment in long‑term
as ≥ 75% intake, all
with no difference
only in the ginkgo
flu like symptoms
tion due to head‑
participants were
Retention/adher‑
nificant improve‑
recall and Stroop
in RAVLT delayed
between groups
of 3.9 (out of 84)
group reporting
aches and sleep
in total, average
Adverse events:
groupmonnieri
Cognition: sig‑
Cognition: sig‑
tablets missed
disturbance
compliant
cognition Results
group
lems
(executive function),
12 weeksWoodcock‑Johnson
Psycho‑educational
Additionally, Stroop
and the WAIS letter
and decision time),
memory, cognitive
digit test of imme‑
irrelevant informa‑
processing speed,
score from RAVLT.
(vocabulary), self‑
(both movement
age and retrieval,
ordered pointing
crystallised abil‑
ability to ignore
inspection time
long‑term stor‑
battery‑revised
spot‑the word
delayed recall,
ity, short‑term
Delayed recall
task assessing
diate working
odd‑man‑out
control and dose DurationMeasures of
tion, the DAT
(fluid ability,
memory
6 weeks of placebo
ofBacopa monnieri
for the treatment
for control group
and 12 weeks
group
Matched placebo (3
extract, 300 mg/day
per meal), N.R = pla‑
tablets per day—1
of 40 mg each—1
Matched placebo
cebo ingredients
Bacopa monnieri
120 mg per day
()Ginkgo biloba
(1 tablet daily),
(3 tablets daily
measure DesignIntervention/
(1 tablet daily)
™Ginkgoforte
N.R = placebo
Standardised
ingredients
per meal)
trial, outcomes at 0,
trial, outcomes at 0
placebo control
placebo control
6 and 12 weeks
and 12 weeks
double‑blind
double‑blind
Randomised,
Randomised,
and names of medi‑
BOMC and no com‑
study location AimStudy populationDiagnosis criteria/
impairment, POMS,
N.R = BOMC cut‑off
cognition measure
POMS, N.R = global
plaints of memory
cal questionnaires
Medical question‑
global cognition
rent medications,
history and cur‑
naire—medical
independently aged
Healthy older adults
also included in this
study however ana‑
females (21 females
participants in each
N.R = range, S.D (27
50 males (26 males
range 55–79 years:
in ginkgo) and 43
65 years or older
54 healthy older
lysed separately
Younger adults
61.7 (± 5.5)m
women) living
=73.5 years,m
in placebo, 24
in placebo, 22
adults (60%
in ginkgo)
group)
ity in healthy elderly
safety and tolerabil‑
(whole plant stand‑
[23]Australia To extend on previ‑
ardised dry extract)
adults on cognitive
ofBacopa monnieri
on cognitive func‑
of 120 mg ginkgo,
tion and its affect,
mance of healthy
mine the efficacy
to benefit perfor‑
in order to deter‑
older and young
ous research
participants
2008[24]U.S.A To evaluate
the effects
outcomes
Burns et al., 2006
Calabrese et al.,
Author and
Table 3(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
nificant improvement
out, > 95% adherence
reports of headaches,
effects were reported
excluded from analy‑
strict task for the pla‑
Cognition: no signifi‑
in neuropsychologic
mon AEs being nau‑
pliance, N.R = adher‑
sis due to non‑com‑
with the most com‑
cant improvements
ence: 3 participants
ence: 3 participants
and flatulence, two
sea/stomach upset
17% of each group
only in list learning
no serious adverse
reported they had
rash and dizziness
for all participants
one AE attributed
from the placebo
Retention/adher‑
Retention/adher‑
from the Ginkgo
Adverse events:
Adverse events:
N.R = minor AEs
group dropped
by participants,
to the product
Cognition: sig‑
they ingested,
group and 8
cebo group
test data
ence
4 monthsBenton visual reten‑
tion, controlled oral
judgement of line
mental screening,
word association,
list learning strict
list learning easy,
and symbol digit
modified mini
orientation,
modalities
68 mg of,Gotu kola
containing 160 mg
daily multivitamin/
based supplement
day with meals. All
(100 mg) and vita‑
also given a once‑
plement (Nutrilite
multimineral sup‑
Matched placebo
participants were
as beta carotene,
of,Ginkgo biloba
180 mg of DHA,
day with meals,
three capsules/
three capsules/
a bioflavonoid
min A (300 IU)
‑Ginkgo biloba
N.R = placebo
concentrate
ingredients
Daily)
Making Test (parts
6 weeksSelective Remind‑
the WAIS‑3 Digit
Symbol‑Coding
ing Test, WMS‑3
(face 1 and face
2 subtests), Trail
A and B), SCWT
subtest
32 oz/day (2 × 16 oz)
Matched placebo
N.R = specific pla‑
cebo ingredients
beverage 32 oz/
containing 27%
cranberry juice
day (2 × 16 oz),
with sucralose
of a beverage
sweetened
trial, outcomes at 0
placebo control
and 4 months
double‑blind
MMSE (24–29)Randomised,
range 65–84 years),
(± 6.0) years, range
tively intact older
73.1 (± 4.8) years
group (21F:21 M
Healthy, cogni‑
placebo group
(15F:21 M, 72.1
adults: ginkgo
65–83 years)
mostasis, and is safe
alters primary hae‑
plement improves
cognitive function
and quality of life,
tively intact older
in healthy, cogni‑
if aGinkgo biloba
containing sup‑
[25]USA To determine
adults
Carlson et al., 2007
trial, outcomes at 0
placebo control
double‑blind
Randomised,
and 6 weeks
no history dementia
neurocognitive
and reporting
or significant
MMSE (≥ 24)
impairment
(± 5.80) (N.R = range,
cranberry group (25
(> 60 years of age):
placebo group (25
Cognitively intact
adults)=69.17m
adults)=69.39m
(± 7.11) years
older adults
sex)
efficacy of cranberry
[27]USA To conduct the first
functioning of cog‑
nitively intact older
known clinical trial
of the short‑term
juice on the neu‑
ropsychologic
adults
Crews et al., 2005
Table 3(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
from the final analysis
compliance reported
as 100% for nearly all
ticipant experienced
participant reported
and VFT across time,
oxidant group com‑
in TMT A and TMT B,
cant improvements
of sinusitis in active
event of hepatitis E
(1 adverse event, 1
due to withdrawal
ticipants excluded
non‑serious event
MoCA and RAVLT Cognition: signifi‑
Adverse events: 1
Retention/adher‑
pared to placebo
could not attend
group and 1 par‑
an acute serious
of exacerbation
study sessions),
and in the anti‑
participants
ence: 2 par‑
TMT, VFT, MMSE,
followed by 8 weeks
intake or interven‑
of what occurred
during this time)
tion or placebo
period (unclear
1‑week run‑in
once daily contain‑
containingBacopa
vegetable stearate
pene, astaxanthin
active substances
Matched placebo
and vitamin B12.
(5 mg) and non‑
Antioxidant mix
ing magnesium
, lyco‑monnieri
1 tablet daily
tablet taken
trial, outcomes at 0
placebo controlled
double‑blind
disorders Randomised,
and 8 weeks
and no neurological
MMSE (≥ 27)
Healthy older adults
(> 60 years of age):
years) N.R = range
=61.88 (± 1.36)m
=62.05 (± 1.55)m
group (28F:12 M,
group (27F:13 M,
antioxidants mix
years), placebo
to 8 weeks of treat‑
ment with an anti‑
oxidant mix com‑
pared to placebo
in healthy older
in the trail mak‑
ing test scores
from baseline
the changes
[39]Italy To evaluate
adults
Crosta et al., 2020
Table 3(continued)
Retention/adherence:
reported, N.R = adher‑
in the ginkgo synergy
from baseline to end‑
oral word association
with ulcerative colitis
for the Ginkgo group
point and in the OPC
Cognition: according
group from baseline
to time in the TMT‑B
joint aches and one
placebo participant
ing, one participant
nificantly increased
in the OPC synergy
shortly after enroll‑
retention numbers
reported insomnia
the Ginkgo group
showed improve‑
on the controlled
group diagnosed
line to 3 months,
ment from base‑
trial‑S scores sig‑
and heightened
Adverse events:
group reported
one participant
energy at night
inconsistently
to 3 months
cognition Results
ence
subtest of the WAIS
the Digit Symbol
A and B, COWA,
and the HVLT‑R
6 monthsMMSE, SCWT,
the TMT Parts
control and dose DurationMeasures of
cebo administration
lets/day to 700 mg/
®1) Ginkgo Synergy
method/type, dose
3) Placebo contain‑
and choline (4 tab‑
(2 capsules/day—
powder, N.R = pla‑
120 mg/day total)
®2) OPC Synergy
lactose and beet
(2 capsules/day)
measure DesignIntervention/
and Catalyn (4
ing cellulose,
tablets/day)
day total)
trial, outcomes at 0,
placebo control
3 and 6 months
double‑blind
Randomised,
study location AimStudy populationDiagnosis criteria/
disorders, not living
SPMSQ, WMS‑Story
A, no AD or related
in a nursing facility
global cognition
MMSE (≥ 23),
cebo group=70.3m
Healthy older adults
®Synergyplus Cata‑
(± 6.7) years, range
(± 8.3) years, range
lyn group=68.5m
with 25F:8 M; OPC
with 24F:7 M; pla‑
®Ginkgo synergy
=67.6 (± 6.3)m
with 21F:12 M
plus Choline
59–83 years,
60–93 years,
58–82 years,
years, range
[29]USA To extend the evalu‑
of nutritional thera‑
placebo‑controlled
pies through a ran‑
clinical trial assess‑
of dietary supple‑
ments’ efficacy
ing a regimen
double‑blind,
ative process
domised,
Lewis et al., 2014
Author and
nificant improvement
compared to placebo
in colour naming task
Colour and Word test
(missing > 20% treat‑
reasons, N.R = overall
over the trial period
reported by partici‑
females completed
to non‑compliance
3/4 due to medical
in the intervention
no adverse effects
not complete due
group across time
the study (40/44),
within the Stroop
ment), remaining
1 participant did
21 males and 19
Adverse events:
Cognition: sig‑
adherence
cognition Results
pants
and visual repro‑
memory 1 and 2
duction 1 and 2
revised, logical
and the WMS‑
control and dose DurationMeasures of
6 weeksSCWT (parts
A and B)
subtests
per day (methylcel‑
lulose), N.R = addi‑
EGBGinkgo biloba
capsules per day).
cebo × 3 capsules
day = 60 mg × 3
measure DesignIntervention/
tional placebo
761 (180 mg/
Matched pla‑
ingredients
trial, outcomes at 0
placebo control
double‑blind
Randomised,
and 6 weeks
of significant neuro‑
AimStudy populationDiagnosis criteria/
cognitive dysfunc‑
global cognition
cognitively intact
tion, considered
and no history
(self‑reported)
MMSE (≥ 24)
healthy older adults,
55–86 years of age:
Cognitively intact
group=68.65m
=67.5 (± 9.23)m
years, placebo
ginkgo group
(± 6.95) years,
24F:24 M
tively short‑term (i.e.
To examine the rela‑
functioning of cog‑
ropsychologic tests
nitively intact per‑
sons over the age
6 weeks) efficacy
on the cognitive
ofGinkgo biloba
extract EGB 761
battery of neu‑
and measures
via a diverse
of 55 years
Table 3(continued)
Mix et al., 2000USA
improved in SRT tasks
from the intervention
group were excluded
in the placebo group
tocol, 5 from the pla‑
ticipants in the inter‑
(missing > 20% treat‑
249/262 participants
testinal, nervous sys‑
tem and respiratory/
completed the pro‑
across both groups.
total of 32 reported
from the study due
ment), N.R = overall
limited to gastroin‑
free recall and rec‑
cebo) significantly
to non‑adherence
One serious event
(compared to pla‑
cebo group and 4
involving delayed
including but not
ognition of visual
allergic reactions,
of an intracranial
Adverse events:
Cognition: par‑
vention group
adherence
material
cognition Results
bleed
6 weeksSRT, WAIS‑3 block
subtests, WMS‑3
Symbol‑Coding
control and dose DurationMeasures of
faces 1 and 2
design, Digit
subtests
tablets × 3 per day,
EGBGinkgo biloba
Matched placebo
tablets per day).
day = 60 mg × 3
measure DesignIntervention/
N.R = placebo
761 (180 mg/
ingredients
trial, outcomes at 0
placebo control
double‑blind
Randomised,
and 6 weeks
nificant neurocogni‑
AimStudy populationDiagnosis criteria/
of dementia or sig‑
global cognition
tive impairment
and no history
(self‑reported)
MMSE (≥ 26)
after withdrawals/
non‑compliance,
adults, ≥ 60 years
=66.97(± 6.12)m
group=68.60m
ticipants in total
years, placebo
ginkgo group
(± 6.96) years,
with 262 par‑
(N.R = range),
in the study,
in the study
intact older
147F:102 M
Cognitively
remained
of age:
chological function‑
To conduct the first
intact older adults
ing of cognitively
on the neuropsy‑
scale clinical trial
ofGinkgo biloba
extract (EG 761)
of the efficacy
known, large‑
Table 3(continued)
Mix et al., 2002USA
total learning and ret‑
occurred significantly
completed the study.
roactive interference
group (side effects
and delayed recall,
Cognition:Bacopa
81/98 participants
frequency, nausea
learning, memory
more in the treat‑
Adverse events: 9
group, 2 placebo
improved verbal
in the treatment
N.R = adherence
and abdominal
increased stool
ment group:
significantly
acquisition
cramps)
cognition Results
(RAVLT)
measured by RAVLT,
ured by the MAC‑Q
and visual memory
CFT and the Reitan
the Rey‑Osterrieth
complaints meas‑
TMT, subjective
control and dose DurationMeasures of
performance
12 weeksAudio‑verbal
Matched placebo
tablet, N.R = dose,
300 mg/day (one
Bacopa monnieri
measure DesignIntervention/
™(BacoMind)
tablet daily).
ingredients
trial, outcomes at 0
placebo control
and 12 weeks
double‑blind
Randomised,
of depression (≤ 12)
study location AimStudy populationDiagnosis criteria/
global cognition
and absence
MMSE (≥ 24)
on HAMD
Healthy older adults
=65(± 7.53) years,m
range 55–86 years
group = 24F:25 M,
group = 28F:21 M
eral population
from the gen‑
placebo
Bacopa
ofBacopa monnieri
Linn. for improve‑
ment of memory
the effectiveness
in healthy older
[32]Australia To investigate
performance
persons
Table 3(continued)
Morgan et al., 2010
Author and
Table 3(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
to endpoint in imme‑
groups from baseline
N.R = specific adher‑
across both groups,
reported by partici‑
nificant differences
for the older adults
between the treat‑
ence: 1 participant
missed dose of 2.2
no adverse effects
recall for the older
Cognition: no sig‑
did not complete
Retention/adher‑
ment or placebo
the study (44/45
diate or delayed
Adverse events:
completed),
an average
group
pants
ence
tests of verbal epi‑
and delayed recall
2 weeksPen and paper
sodic memory
immediate
L.) ethanolofficinalis
5 ml of SRM (Salvia
(L.)), 1% Lyles Black
N.R = specific time
fresh sweet cicely
marinus officinalis
diluted in water)
L.,officinalisRos-
Treacle and 1 g/
(Myrrhis odorata
of day for dose,
extract or 5 ml
on smell, taste
twice per day,
ml 45% EtOH,
placebo (50%
L. andMelissa
or matched
trial, outcomes at 0
placebo control
double‑blind
Randomised,
and 2 weeks
of cognitive impair‑
N.R = global cogni‑
ment or dementia,
clinical diagnosis
No reported cur‑
rent or previous
tion measure
Healthy older adults
=61 (± 9.26) yearsm
38F:6 M total (older
tion group and 2
groups: younger
bers: 4 males = 2
in the interven‑
14 females = 10
and 4 placebo)
in intervention
(divided into 2
in the placebo
(43–62), older
group num‑
(63–80))
group
medicines on verbal
ditional European
and Melissa—tra‑
of sage, rosemary
of a combination
healthy subjects
time the effects
recall in normal
[34]UK To evaluate
for the first
Perry et al., 2018
Table 3(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
dosage, N.R = specific
not monitored in The
ticipants completing
and 9 in the placebo
ment groups on any
ply with medication
in the ginkgo group
group did not com‑
in non‑completion,
nificant differences
nificant differences
between the treat‑
outcome measure
withdrew consent
Cognition: no sig‑
Retention/adher‑
or between time
between groups
with 88% of par‑
Adverse events:
the trial, no sig‑
ence: 4 ginkgo
and 7 placebo
7 participants
adherence
points
study
test, Boston naming
of the WMS‑revised,
(WMS‑R) controlled
and mental control
tion, concentration
scale of the WAIS‑
Memory subscale
utilising the CVLT,
digit symbol sub‑
duction subscale,
revised), the digit
category fluency
the Visual Repro‑
language tested
memory, atten‑
and expressive
span (WMS‑R)
the Logical
6 weeksLearning,
test
times per day. States
is a tablet, placebo
matched placebo
Ginkgo (Ginkoba)
1 capsule 3 × per
day. Note: active
(lactose gelatin),
40 mg 1 tab‑
is a capsule
let, taken 3
trial, outcomes at 0
placebo control
double‑blind
good health Randomised,
and 6 weeks
and in generally
MMSE (> 26)
years, with 65F:46 M,
and ages calculated
years with 63F:45 M
dwelling volunteer
numbers, F:M ratio
after withdrawals)
aged between 60
older adults (total
of 132F:98 M, 115
in each group),
to intervention
=68.7 (± 4.7)m
=69.9 (± 5.4)m
placebo group
Ginkgo group
Community‑
and 82 years
(assignment
tests and subjective
neuropsychological
improves memory
whether ginkgo
in elderly adults
as measured
by objective
[36]USA To evaluate
ratings
Solomon et al., 2002
Table 3(continued)
cognition Results
control and dose DurationMeasures of
measure DesignIntervention/
study location AimStudy populationDiagnosis criteria/
global cognition
Author and
cance for ages 60–81)
Retention/adherence:
tion period (semantic
Adverse events: none
(Japanese version) Cognition: significant
drew from the study
fluency), total score
independent of sex
reported by partici‑
3 participants with‑
approached signifi‑
endpoint interven‑
sub‑group analysis
increase in RBANS
between baseline
but age‑depend‑
and the 12‑week
N.R = adherence
for 47–81 years,
ent (significant
(31 originally),
pants
RBANS, MMSE‑J
12 weeks intake,
washout period
with a 6‑week
the crossover
separating
period
was matched in col‑
are taken, if placebo
and white beeswax,
15 containing 8 mg
Diosgenin‑rich yam
vitamin E derivative
vitamin E derivative
and white beeswax
our, smell and taste
containing olive oil
diosgenin, 672 mg
are taken. Placebo
(672 mg), glycerol
N.R = when doses
N.R = when doses
extract diopower
olive oil, glycerol
(2 capsules/day)
fatty acid ester,
fatty acid ester,
day = 50 mg),
(2 capsules/
to active
trial, outcomes at 0,
Randomised, dou‑
ble‑blind placebo
control, crossover
12 and 30 weeks
physical and mental
health, no diagnosis
of AD or related dis‑
orders, N.R = global
cognition measure
Overall good
(subgroup for analy‑
numbers calculated
(16F:12 M)46.5m
60–81 years) (ages
range 20–81 years
after withdrawals)
sis of older adults
and participant
(± 18.67) years,
Healthy adults
yam extract on cog‑
of a diosgenin‑rich
ment in healthy
nitive enhance‑
[40]Japan Investigate
volunteers
the effects
Tohda et al., 2017
ADAEBOMCCFTCOWACVLTDATAlzheimer’s disease,Adverse event,Blessed Orientation Memory Concentration test,Complex Figure Test,Controlled Oral Word Association test,California Verbal Learning Test,Divided
HAMDHVLT-RMAC-QMMSEMoCAAttention Task,Hamilton Rating Scale for Depression,Hopkins Verbal Learning Test-Revised,Memory Complaint Questionnaire,Mini-Mental State Examination,Montreal Cognitive
N.RPOMSRAVLTRBANSSCWTAssessment,Not reported,Profile of Mood States,Rey Auditory Verbal Learning Test,Repeatable Battery for the Assessment of Neuropsychological Status,Stroop Colour and Word Test,
SPMSQSRTTMTVFTWAISWMSShort Portable Mental Status Questionnaire,Selective Reminding Test,Trail Making Test,Verbal Fluency Test,Wechsler Adult Intelligence Scale,Weschler Memory Scale
38], compared to 1136 from the twelve studies in older adults without SCI [23–25, 27, 29–32, 34, 36, 39, 40].
Eligibility criteria and global cognition measures
All twenty-one studies utilised cognitive scales or tests
- [20–22, 25–33, 35, 36, 38, 39], medical questionnaires [23, 37], self-reports of cognitive function [22, 24, 26,
- 27, 29–31, 34, 40] or clinical questionnaires [23, 24, 32, 38], to determine the eligibility for their respective study. Nineteen of these studies utilised a validated measure to test cognitive functioning [20–33, 35, 36, 38–40]. Fifteen out of twenty-one studies utilised the Mini-Mental State Exam (MMSE), as a measure of global cognition [20–22, 25–33, 35, 36, 38, 39]. The MMSE cut-off score for participant inclusion varied between studies and populations. For SCI studies, one utilised a cut-off of≥ 20 [22], another a range of 20–27 [26], one with≤ 24 [35], four studies with a cut-off of≥ 24 [21, 28, 33, 38] and one of≥ 25 [20]. For studies on older adults without SCI, one reported a cut-off of≥ 23 [29], one with a range of 24–29 [25], three with a cut-off of≥ 24 [27, 30, 32], one≥ 26 [31] and one each > 26 [36] and≥ 27 [39]. Overall, studies with an SCI population reported lower cut-off scores and ranges for participant inclusion.
Other scales included the Blessed Orientation Memory Scale (BOMC) [24], Weschler Memory Scale (WMS) [28, 29, 35], Clinical Dementia Rating (CDR) Scale [20], Memory Complaint Questionnaire (MAC-Q) [28] and Short Portable Mental Status Questionnaire (SPMSQ) [29]. Overall, four studies utilised a global cognition measure as their primary or secondary outcome measure, with three of these using the MMSE [26, 29, 39], and one the WMS (logical subset score of < 6) [35].
Two of the remaining four studies utilised a herbal combination via a liquid solution (Ginkgo biloba, alcohol/water solution [22] and SRM [Salvia officinalis L., Rosmarinus officinalis L. and Melissa officinalis L.]) [34], one study utilised a nutrient based juice (sweetened cranberry juice) [27], and one study used an oral capsule containing a type of fungus (Tremella fuciformis) [20]. Twenty out of twenty-one studies reported a method of administration [20–34, 36–40] with ten administering the intervention orally via capsule [20, 21, 25, 26, 28–30,
- 33, 37, 40], seven utilised tablets [23, 24, 31, 32, 36, 38, 39] and three a liquid solution [22, 27, 34].
Intervention and control type
Seventeen out of twenty-one studies used a herbal supplement [21, 23–26, 28–33, 35–40] with most studies utilising a form of Ginkgo biloba [23, 25, 29–31, 36–38] or Bacopa monnieri as a primary ingredient in their intervention [21, 24, 26, 32, 35, 39]. Two of these seventeen studies used a combination formula (one containing Ginkgo biloba and 45 other herbs, minerals and vitamins specifically made for women [38]) (the other containing Bacopa monnieri, lycopene, astaxanthin and vitamin B12 [39]). In addition, one study each used a spearmint extract (Mentha spicata L.) [28], a standardised ginseng extract (G115) [33] and a diosgenin-rich yam extract (diopower 15) containing vitamins, oils and beeswax [40], and one used a nutritional supplement (OPC Synergy® plus Catalyn) containing buckwheat, teas, and fruit and vegetable extracts, as a secondary intervention [29].
Fig. 2 Risk of bias domains for older adults with SCI (parallel studies)
Retention and adherence
Nineteen of the twenty-one studies consistently reported retention, with an average retention rate of 92% across the studies [20–26, 28, 30–38, 38–40]. Comparatively, only 7/21 studies specifically reported treatment adherence, with an overall average of 93% across both intervention and placebo usage [20, 22, 24, 26, 28, 33, 38, 38].
Risk of bias within and across studies
Figures 2 and 3 (parallel studies) and Fig. 4 (cross-over study) provide a summary of each of the risk of bias domains, and an overall risk of bias assessment, for each of the twenty-one included studies. Green circles indicate that the domain or study has been evaluated as low risk, yellow as having some concerns and red as high risk.
Each article was assessed in terms of randomisation, intended interventions (effect of assignment and effect of adhering to the intervention), missing outcome data, measurement of outcomes and selection in reported results. In terms of the overall risk of bias assessment, only one study was deemed as low risk [26], some concerns were found for 6/21 studies [20, 24, 27, 28, 33, 39] and the remaining fourteen were deemed to be high risk
- [21–23, 25, 29–32, 34–38, 40]. Between populations, three SCI studies were assessed as having some concerns
[20, 28, 33], five were deemed high risk [21, 22, 35, 37, 38] and one was deemed low risk [26]. For the non-SCI studies, three were assessed as having some methodological concerns [24, 27, 39], and the remaining nine were deemed as high risk [23, 25, 29–32, 34, 36, 40].
Despite all twenty-one studies stating the method of intervention assignment was randomised, only fourteen sufficiently detailed the randomisation process and were deemed as low risk [21–23, 25–27, 29–32, 36–38, 40]. Intended interventions (effect of assignment to interventions) were adequately reported in five studies (low risk) [20, 26, 28, 29, 36], nine were assessed as high risk [21– 23, 25, 30–32, 38, 40] and the remaining seven have some concerns [24, 27, 33–35, 37, 39]. Thirteen studies were assessed as low risk for reporting on intended interventions (effect of adhering to the intervention) [20, 22–24, 26–31, 33, 38, 39] and eight as high risk [24, 29, 32, 34– 37, 40].
In terms of missing outcome data (domain three), 17/21 studies were evaluated as low risk [20–22, 24, 26– 31, 33–37, 39, 40], and the remaining four were assessed as high risk [23, 25, 32, 38]. Within domain 4 (measurement of outcomes), 18/21 studies were deemed as low risk [20–33, 36–39], and the remaining three were as high risk [34, 35, 40]. Eighteen out of twenty-one studies
- Fig. 3 Risk of bias domains for older adults without SCI (parallel studies)
- Fig. 4 Risk of bias domains for older adults without SCI (cross‑over study)
were assessed as low risk for selection in reported results (domain 5) [20–22, 24–29, 31–39], with some concerns for only three studies [23, 30, 40]. In terms of bias arising from period or carryover effects (domain S) in the crossover study, this was deemed as low risk [40].
Study results
Results for all twenty-one studies are outlined below including intervention efficacy on cognitive function, adverse events and risk of bias.
Intervention efficacy in low risk of bias study
Only one study was deemed to be low risk for all domains, in terms of the overall methodological assessment. This 2017 randomised, double-blind placebocontrolled trial was conducted by Cicero and colleagues [26]. Participants were 30 older adults with self-perceived cognitive decline and ingested either a Bacopa monnieri formulation or a placebo capsule for 8 weeks; MMSE was measured at each time point. In terms of intervention efficacy, a significant increase in MMSE score was
found from baseline to endpoint in the treatment arm. Furthermore, a significant increase in score was also found in the treatment group at the endpoint, compared to placebo, demonstrating a significant improvement in cognitive function for the intervention group across time and between groups [26]. Only one adverse event was reported an aftertaste from active product intake.
Intervention efficacy in remaining SCI studies
Across time, a significant improvement in at least one cognitive outcome for participants in the intervention group (compared to placebo) was found in 6/8 of the remaining SCI studies [20, 21, 28, 33, 35, 38]. Improvements were mostly found in the areas of memory (working, spatial, short-term, retention and logical) [20, 28, 33, 35, 38] and executive functioning [20, 21, 35]. Three of the eight studies utilised a capsule containing a herbal extract: one contained Bacopa monnieri [21], one spearmint extract (Mentha spicata L.) [28] and one standardised ginseng extract [33]. One study used a combination supplement (tablet), containing 46 herbs, vitamins and minerals (mainly consisting of Ginkgo biloba, Silybum marianum dry fruit (St. Mary’s thistle) and Vitis vinifera dry seed (grape seed)) [38]. An additional study utilised a capsule containing Tremella fuciformis (a type of fungus) [20], and the last study did not specify an administration method but used a standardised extract of Bacopa monnieri [35].
Of these six studies, mild to moderate adverse events were reported in three of them [28, 35, 38]. Knee pain, myalgia, headaches and heartburn were reported in the study conducted by Herrlinger and colleagues utilising Mentha spicata L. as their intervention [28]. These adverse events were reported for both the treatment and placebo groups; however, heartburn experienced by a participant in the 600 mg/day Mentha spicata L. group was deemed as ‘probably related’, compared to all other events deemed as ‘not related’ [28]. One participant withdrew due to maculopapular rashes in the intervention group (Bacopa monnieri), in the study conducted by Raghav and colleagues [35]. Two participants withdrew from the study conducted by Macpherson and colleagues using a combination formula containing Ginkgo biloba (intervention) [38]. One participant withdrew due to nausea and vomiting in the intervention group and one in the placebo group due to a mild rash [38].
In relation to the risk of methodological bias for all six SCI studies with an improvement in cognitive functioning, two of the studies utilising Bacopa monnieri [21, 35] and one using Ginkgo biloba (combination supplement) [38] were deemed as being high-risk. The remaining three were assessed as having some concerns in terms of methodological reporting [20, 28, 33].
For the two studies that did not find an improvement in cognitive functioning between groups (intervention cf. placebo) or across time, both reported adverse events with the use of Ginkgo biloba alcohol/water extract (drops) [22] and a herbal/dietary supplement also containing Ginkgo biloba [37]. Gastrointestinal upset was reported as the main adverse reaction for both studies [22, 37] with dizziness, headaches and sleep disturbance also reported in the Ginkgo biloba alcohol/water extract study [22]. In total, Brautigam and colleagues reported adverse events for 25 participants across both the placebo and intervention groups [22]. In terms of the second study, it is not known whether the two participants who reported adverse events were receiving the intervention or placebo [37]. Overall, both studies were deemed as high risk in terms of the methodological risk of bias assessment.
Intervention efficacy in non‑SCI studies
Overall, 7/12 non-SCI studies reported a significant improvement in cognitive functioning in the intervention group (compared to placebo), across time [23, 24, 30–32, 39, 40]. The most common improvements were in memory (long-term storage, retrieval, delayed recall, recognition) [23, 24, 31, 32], executive functioning [24, 30, 39, 40] and language [30, 39, 40]. Two studies used a form of Ginkgo biloba capsule [23, 30]; one used a Ginkgo biloba tablet [31]; two used a form of Bacopa monnieri tablet [24, 32]; one used an antioxidant combination formula (tablet) containing Bacopa monnieri, lycopene, astaxanthin and vitamin B12 [39]; and one a diosgenin-rich yam extract capsule [40]. An additional study reported a significant improvement in executive functioning across time, using a combined herbal and nutritional supplement containing Ginkgo (Ginkgo Synergy® plus Choline) [29]. A significant improvement in verbal fluency was also found in the secondary intervention group (across time, compared to the Ginkgo and placebo groups) using OPC Synergy®, a dietary supplement (plus Catalyn) [29]. A further study using a Ginkgo biloba-based supplement found a significant improvement in a list learning strict task in the placebo group only, across time [25].
Seven of the nine total studies were deemed as having a high methodological risk of bias [23, 25, 29–32, 40], with the remaining two (using a form of Bacopa monnieri) having some concerns [24, 39]. Five of the nine studies reported adverse events, in both the placebo and intervention groups (Ginkgo or Bacopa monnieri interventions) [24, 25, 29, 31, 32]. An additional two studies reported adverse events only occurring in the intervention group using a Ginkgo biloba capsule [23] or an antioxidant combination formula containing Bacopa monnieri [39]. The most common events reported across
6/7 studies were gastrointestinal issues (including nausea, abdominal cramps, digestive problems) [24, 25, 31, 32] and sleep disturbance, with the use of Ginkgo biloba [23] or placebo [29]. Exacerbation of sinusitis (n= 1) and a serious but short-term event of hepatitis E (n= 1) were reported as non-treatment-related adverse events in the remaining study [39].
For the three remaining non-SCI studies, all reported no significant improvements across time in cognitive functioning (for both intervention and placebo groups), nor between groups [27, 34, 36]. One study used a Ginkgo tablet [36], one used a sweetened cranberry juice [27] and the other used a liquid solution of SRM Salvia officinalis L., Rosmarinus officinalis L. and Melissa officinalis L. [34].
Adverse events were monitored in two of the three studies; however, no events were reported in one (SRM solution) [34], and the other study did not report serious events or document mild/minor events (sweetened cranberry juice intervention) [27]. Two of the three studies were deemed as having a high methodological risk of bias [34, 36], and the remaining one had some concerns [27].
Discussion
Overall, twenty-one studies were identified for inclusion in this review, nine with an SCI population [20–22, 26,
- 28, 33, 35, 37, 38] and twelve studies utilising older adults without SCI [23–25, 27, 29–32, 34, 36, 39]. Outcomes were mainly positive, with 14/21 studies overall reporting improvements in at least one area of cognitive functioning across time, in the intervention group (compared to placebo) [20, 21, 23, 24, 26, 28, 30–33, 35, 38–40]. Overall, only one study (using an SCI population) was assessed as having a low methodological risk of reporting, conduct and quality of trial design bias [26]. Due to the heterogeneous nature of eligible studies (including cognitive measures and interventions used and the type of data analysis and reporting conducted) and the large number of studies (14/21) with a high methodological risk of bias [21–23, 25, 29–32, 34–38, 40], a certainty of evidence analysis (GRADE) was not conducted.
Extent of literature using herbal and nutritional medicines for older adults with and without SCI
Despite the growing interest in the prevention and treatment of cognitive decline in older adults [8, 9], review search outcomes were not reflective of this interest, given that most of the articles eligible for this review were conducted prior to 2018. Editorials, book chapters, reviews and opinion articles seem to be more common formats of evidence, compared to research using the ‘gold standard’ method, randomised control trials (RCTs) [41, 42] (Fig. 1).
Measurement of cognitive change was not common in the literature; rather, relevant population studies focussed on biochemistry or progression to MCI or dementia. This was an unexpected outcome during the records search. Cognitive testing and assessment are generally affordable and accessible methods of providing insights into an individual’s current cognitive functioning and any decline over time [43]. However, there are currently no recommendations for specific primary or secondary outcome measures of cognition to determine a clinically significant improvement in cognitive function [44]. It has recently been recommended that composite outcomes including the monitoring of dementia risk factors alongside changes in cognition may be advantageous in preclinical dementia [44].
For cognitive outcome measures, an effect size of 0.40 has been reported as a clinically meaningful improvement for cognitive training interventions in healthy older adults and those with MCI or dementia [45]. This could be applied in studies utilising cognitive outcomes in people with SCI to determine whether a change in cognitive function is clinically meaningful, particularly in light of potential ceiling effects in this relatively unimpaired group. Future research should strive to investigate appropriate cognitive measures to detect a clinically significant change in SCI and implement gold standard, high-quality research methods to produce informative and translational outcomes.
Study characteristics
In terms of participant characteristics, across the twentyone studies, there were more females (61%) compared to male participants. It is difficult to ascertain the true difference in the prevalence of SCI between the sexes, as a larger number of females (rather than males) are participating in these studies. Furthermore, inconsistencies in reporting prevalence between the sexes are typical in this field, again making it hard to determine whether SCI affects more females or males [2]. However, research within the area of cognitive decline suggests that females have a greater cognitive reserve but have a faster rate of cognitive decline (particularly, in the areas of global cognition and executive function) compared to males [46]. This outcome has been confirmed in dementia research. Dementia is reported to be the leading cause of death in women, with twice as many females compared to males being affected by the disease [47]. Further SCI prevalence research needs to be conducted to determine the true prevalence of SCI, between the sexes.
A high number of older adults reporting SCI are within the 60–64 year age range [2], compared to studies included in this review that saw an overall average age of
65 years for participants. Sex and age outcomes derived from this review highlight the importance of finding a way to address low research participation in males and monitoring the faster rate of female decline.
In terms of participant retention and adherence to treatment, these were both surprisingly high across the studies at an average of 92% and 93%, respectively, despite the literature suggesting these figures are quite difficult to achieve [48]. These outcomes should be considered with caution due to the subpar methodologies used to treat missing values.
Study methodologies
Reflective on previous research, the eligibility criteria for participation across the SCI and non-SCI studies were inconsistent [8, 9], with varying scales, tests and questionnaires used, particularly for the MMSE [20–22, 25– 33, 35, 36, 38, 39]. Research investigating the diagnostic accuracy (sensitivity, specificity, positive and negative predictive power) of MMSE cut-off scores in detecting cognitive dysfunction found that scores of≤ 26 showed optimal sensitivity and specificity balance, with a correct classification of MCI and dementia in older adults to be 90% [49]. The varying MMSE cut-offs used here (≥ 20, > 25 and≤ 24) may have incorrectly classified participants (with and without SCI), potentially impacting the study outcomes. This interpretation is further supported by the identification of all eligible studies in this review not using a combination of self-report cognitive concerns (in line with the definition of SCI), cognitive scales (such as the MMSE), a general health questionnaire (including non-diagnosis of MCI or dementia) and screening of mental health conditions. Reliability on only one or two measures for classification of an impairment (or no impairment) is problematic and certainly requires future attention within this area of clinical practice and research.
An additional concern regarding the methodologies of accepted studies in this review is the large number of those deemed as having a high risk of bias, particularly within the bias due to assignment and adherence domains [21–23, 25, 29–32, 34–38, 40]. Either intention-to-treat (ITT) or modified intention-to-treat (mITT) approaches were not employed for participants with missing outcomes or outlier data, with participants being excluded completely from the analysis despite being randomised. Future studies in this field should consider using appropriate analysis to treat missing or outlier data, for postrandomisation outcomes as detailed above. The blinding of participants and other individuals involved in the trial was also identified as a concern. However, it is difficult to
ascertain whether it was in fact the blinding process itself that was not conducted appropriately in these studies or if it was simply not reported sufficiently according to the ROB assessment standards. Future studies should look to adopting greater transparency and accuracy in the process (specifically stating who was blinded and how), as this would go a long way in demonstrating non-biassed outcomes.
Intervention efficacy on cognitive function and safety
Despite the majority of studies reporting positive results [20, 21, 23, 24, 26, 28, 30–33, 35, 38–40], a large number of these were deemed as having an overall high methodological risk of bias [21, 23, 30–32, 35, 38, 40]. These methodological concerns unfortunately do not assist in determining the true efficacy of herbal and nutritional medicines on cognitive functioning for older adults with and without SCI. This is particularly the case for Ginkgo biloba [23, 25, 29–31, 36–38] and Bacopa monnieri [21, 24, 26, 32, 35, 39], given how common they were as interventions across the accepted studies of this review.
The efficacy of Ginkgo biloba has been consistently unclear across the spectrum of cognitive decline. An earlier review investigating RCTs using Ginkgo biloba for the treatment of dementia [50] highlighted the concerns around the low quality of studies available, namely to do with utilisation of unsatisfactory methods. However, on a positive note, adverse effects found with the use of Ginkgo biloba (across the accepted studies in this review) appeared to be consistent with those reported with the use of a placebo [23, 29, 31, 38], indicating that Ginkgo biloba may be comparable in terms of safety with placebo intake. These results are in line with what has been found previously in a dementia population [50].
Strengths and limitations of the review
This review had several strengths. A broad and extensive literature search was conducted (in accordance with the aims and PICOS criteria of the review), comprehensively summarising the overall current state of the field. The lack of high-quality research has been addressed, highlighting the specific aspects which require improvement in future studies. The concerns surrounding the classification of SCI and the disparities between current research outcomes and clinical statistics have been presented.
There were a number of limitations to this review. First, the establishment of article alerts from 2018 until the completion of the review meant that despite the authors’ best efforts to monitor the addition of newly published research in each database, it is acknowledged that alerts
may not have been the most appropriate way to capture all potential studies for inclusion. Furthermore, a metaanalysis was not feasible due to the inconsistent classification of SCI and non-SCI samples and the varying cognitive testing measures. The infancy of this area of research (despite broad interest from the general public) makes it difficult to conduct such an analysis at this time. The population was difficult to define due to the inherent heterogeneity of definitions and lack of consensus within the field, particularly with reference to the age range selected (despite being guided by the US CDC’s definition), and the terminology and language used (e.g. person-centred terminology). It is also acknowledged that the current review did not take into consideration the varying terminology utilised to classify ‘older adults’. The exclusion of non-English language studies, the initial article screening conducted by one reviewer and the search strategy developed in consultation with only one librarian were further limitations.
Recommendations for future research
First and foremost, an increased understanding and awareness of the features and characteristics of SCI needs to occur [8, 9]. This should be considered in collaboration with the difference between the presentation of older adults without SCI and those with MCI, in line with the cognitive decline continuum [2]. Future research should aim at clarifying the characteristics, classification measures and features of SCI to allow for more homogeneous sample classification. Overall, by better understanding of SCI, this may provide greater support for outcomes in high-quality efficacy studies utilising herbal and nutritional medicines as a means of managing selfperceived (or subtle) cognitive decline and, ideally, lowering dementia risk or facilitating the secondary prevention of dementia.
The development of a standardised outcome measure package (including cognitive testing, medical questionnaires, self-reports and mental health questionnaires) for use in SCI clinical trials would be the next step in moving the field forward. Increased accuracy in the differentiation between healthy older adults (without SCI) and those with SCI would assist in determining whether herbal and nutritional medicines have a positive effect on cognitive outcomes for this population.
Conclusions
Whilst most studies deemed eligible for inclusion in the review found positive results (particularly, those that used Ginkgo biloba or Bacopa monnieri), these outcomes need to be considered with caution, due to the high risk of methodological bias found. The literature in
this area is in its infancy, with concerns around population and intervention heterogeneity evident. The use of supplements for cognition by older people is an area that attracts much interest from the community, yet our review shows that high-quality research on efficacy and safety is somewhat lagging.
This review has provided an insight into the current state and quality of the literature on the safety and efficacy of cognitive function of herbal and nutritional medicines in older adults with and without SCI.
Acknowledgements Not applicable.
Authors’ contributions AEC and GZS contributed to the conceptualisation and design of the review. AEC took the lead in drafting the review paper. GZS provided guidance throughout. All authors provided critical feedback and reviewed the final manuscript.
Funding AEC’s contribution was supported by PhD scholarship from FIT‑BioCeuticals Pty Ltd. and NICM Health Research Institute, Western Sydney University. GZS’s contribution was supported by funding from a National Health and Medical Research Council (NHMRC)‑Australian Research Council (ARC) Dementia Research Development Fellowship (APP1102532) and an NHMRC Investigator Grant (APP1195709). The funders played no role in this study.
Availability of data and materials Not applicable.
Figures
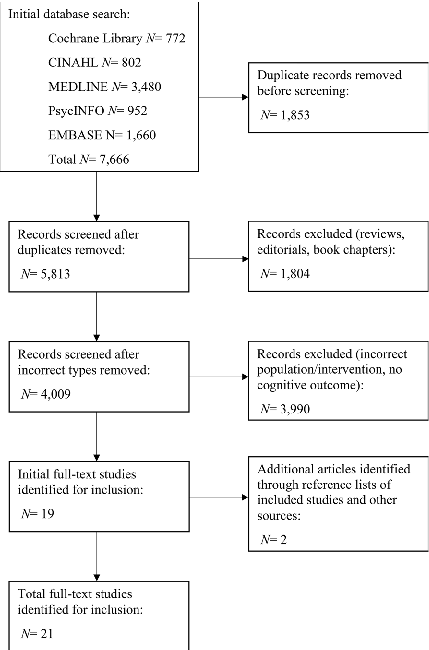
Figure 6
Conceptual diagram summarizing the relationship between a systematic review of the safety and efficacy on cognitive function of herbal and nutritional medicines in older adults with and without subjective cognitive impairment and the biological processes described in this research.
diagram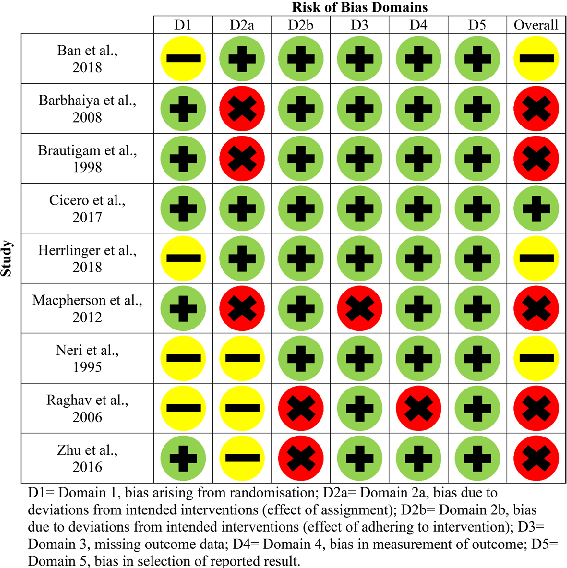
Figure 7
Visual summary of the pathways and interactions relevant to a systematic review of the safety and efficacy on cognitive function of herbal and nutritional medicines in older adults with and without subjective cognitive impairment, as discussed in the context of BACKGROUND: Subjective cognitive impairment (SCI) substantially increases dementia risk and is often conceptualised a.
diagram
Figure 8
Schematic representation highlighting the mechanisms underlying a systematic review of the safety and efficacy on cognitive function of herbal and nutritional medicines in older adults with and without subjective cognitive impairment and their potential therapeutic implications.
diagramUsed In Evidence Reviews
Similar Papers
Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie · 2020
Isorhamnetin: A review of pharmacological effects.
Archives of physical medicine and rehabilitation · 2000
Ginkgo biloba extract: mechanisms and clinical indications.
The Cochrane database of systematic reviews · 2007
Ginkgo biloba for cognitive impairment and dementia.
JAMA · 2009
Ginkgo biloba for preventing cognitive decline in older adults: a randomized trial.
The Cochrane database of systematic reviews · 2009
Ginkgo biloba for cognitive impairment and dementia.
Clinical pharmacokinetics · 2013