Zinc Hình ảnh
23 hình ảnh từ nghiên cứu có bình duyệt
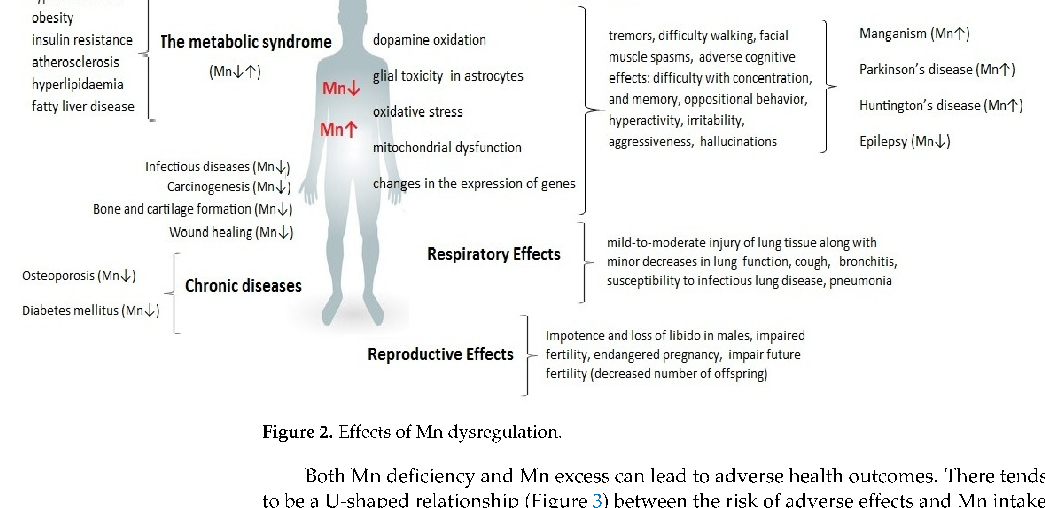
Neurological consequences of chronic manganese exposure are summarized, including cognitive impairment, motor dysfunction, and emotional disturbances. The symptoms collectively termed manganism share clinical features with Parkinson's disease.
Consequences of Disturbing Manganese Homeostasis.
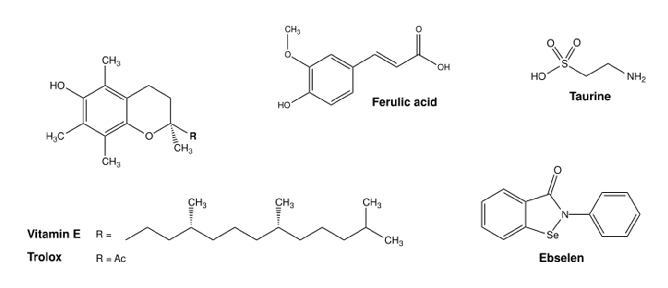
Manganese's dual role as both essential nutrient and potential toxin is explored through its effects on enzymatic systems. Superoxide dismutase, arginase, and glutamine synthetase all require manganese as a cofactor for normal function.
Consequences of Disturbing Manganese Homeostasis.
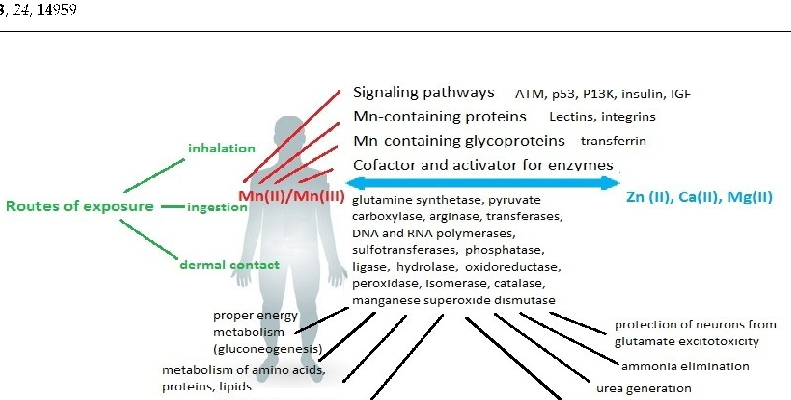
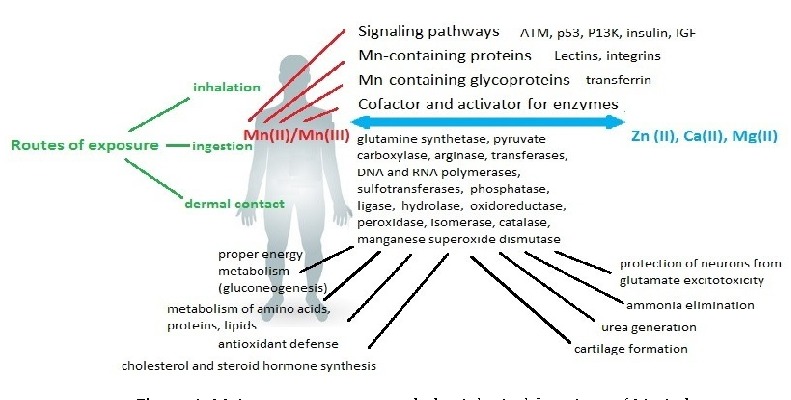
A comprehensive diagram maps the systemic effects of manganese dysregulation across multiple organ systems. Both deficiency and excess disrupt metabolic processes, with the nervous system being particularly vulnerable to manganese imbalance.
Consequences of Disturbing Manganese Homeostasis.
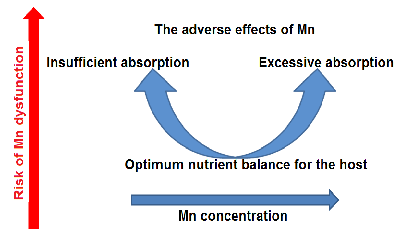
The U-shaped dose-response curve for manganese illustrates that both deficiency and excess are associated with adverse health outcomes. An optimal range exists where manganese fulfills its essential cofactor roles without triggering neurotoxic or systemic damage.
Consequences of Disturbing Manganese Homeostasis.
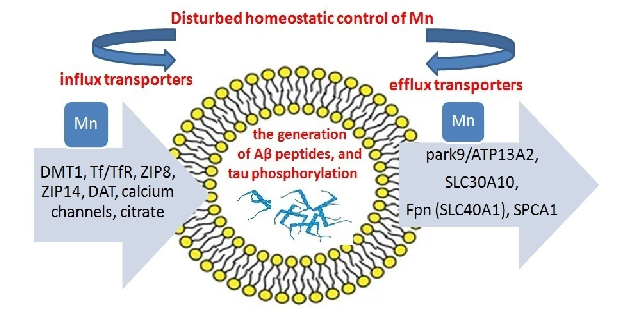
A model of manganese transport imbalance depicts the interplay between uptake, distribution, and excretion mechanisms. Disruption of transporters such as SLC30A10 and SLC39A14 can lead to pathological manganese accumulation in target tissues.
Consequences of Disturbing Manganese Homeostasis.
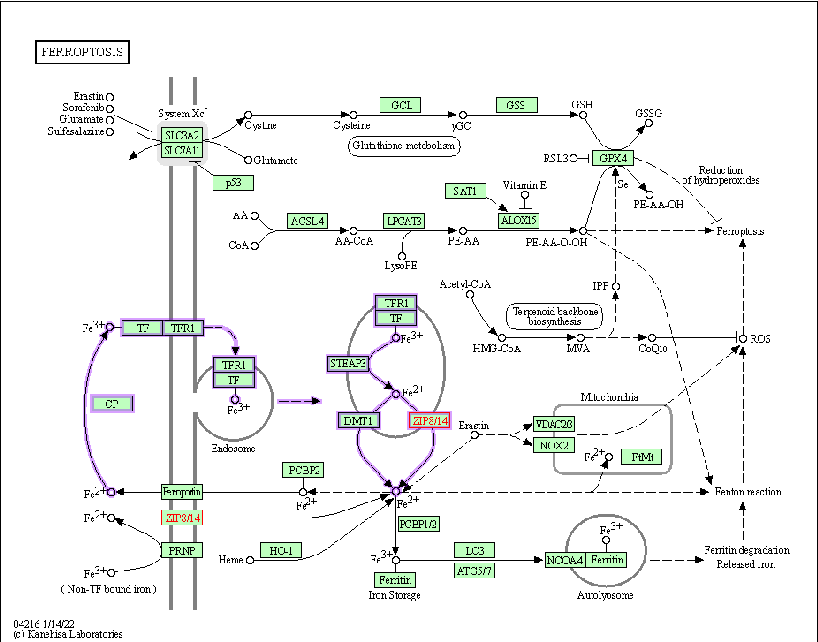
Supplementary or concluding data on manganese homeostasis consequences are presented. The review emphasizes that maintaining manganese within a narrow physiological range is critical for preventing both deficiency-related enzyme dysfunction and toxicity-related neurodegeneration.
Consequences of Disturbing Manganese Homeostasis.
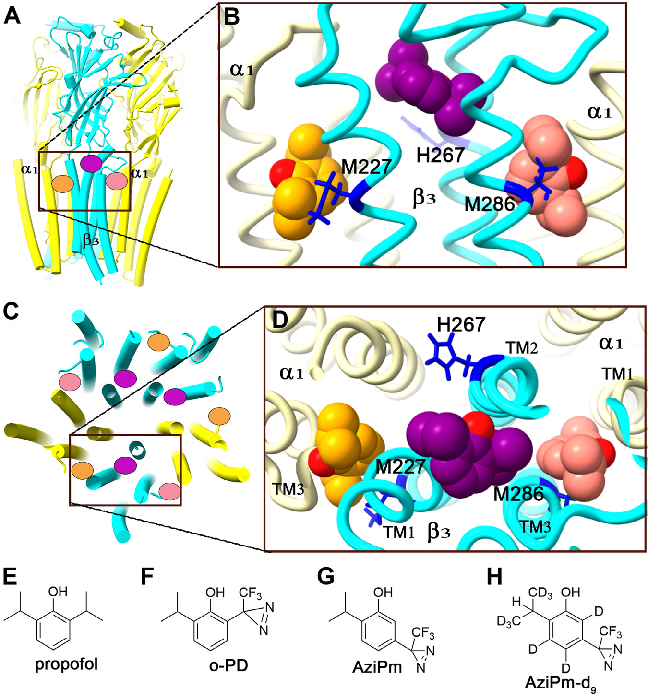
Electrophysiology or binding affinity data for propofol at different GABAA receptor binding sites, comparing potency across the three identified site classes.
Three classes of propofol binding sites on GABAA receptors.
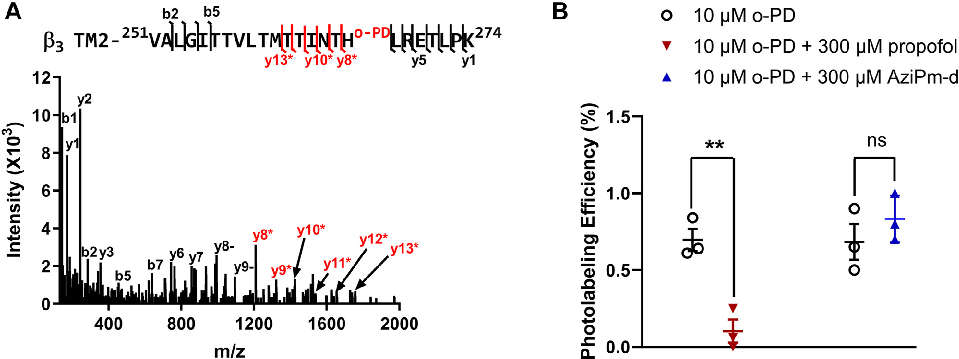
Mutagenesis or competition binding data supporting the classification of three distinct propofol binding site classes on GABAA receptors.
Three classes of propofol binding sites on GABAA receptors.
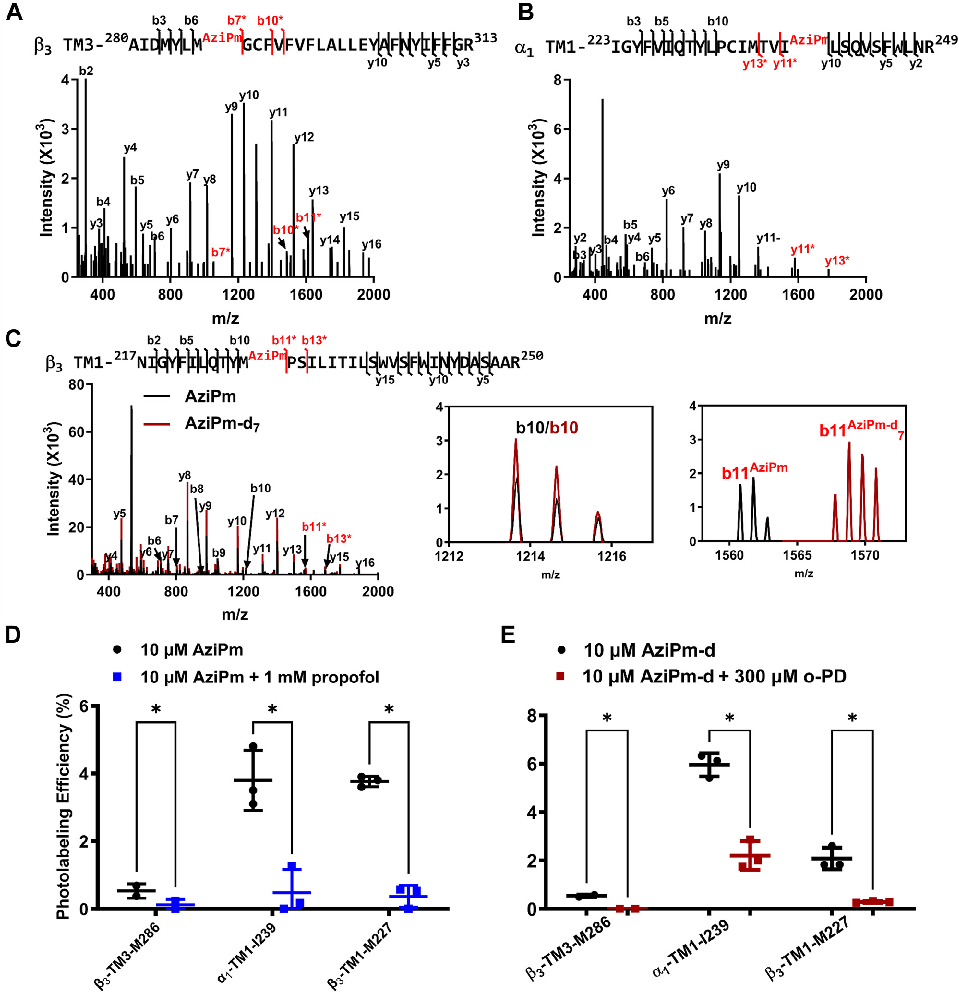
Structure-activity relationship analysis of propofol analogs at the different GABAA receptor binding sites, informing anesthetic drug design.
Three classes of propofol binding sites on GABAA receptors.
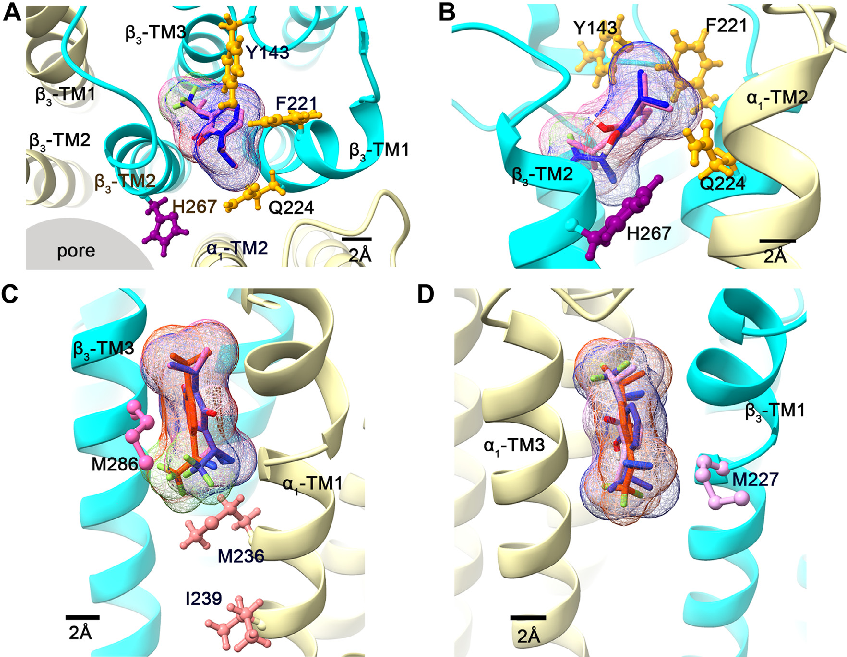
Molecular dynamics simulation or computational modeling of propofol interactions with GABAA receptor transmembrane domains.
Three classes of propofol binding sites on GABAA receptors.
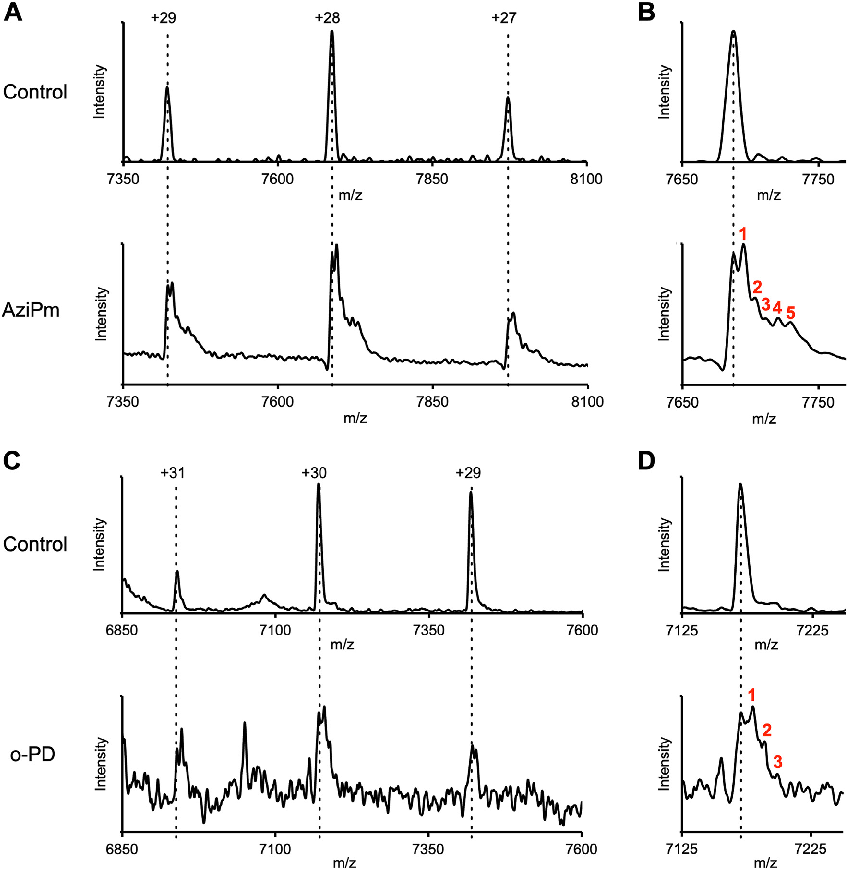
Comparative analysis of propofol binding site occupancy and functional consequences for GABAA receptor channel gating and anesthetic efficacy.
Three classes of propofol binding sites on GABAA receptors.
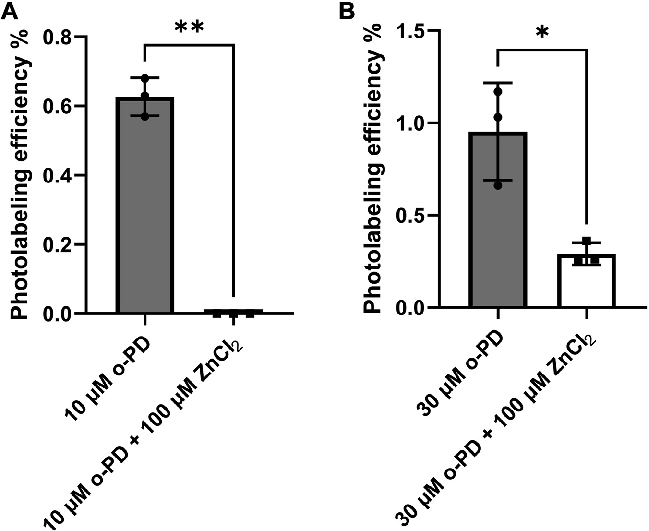
Supplementary structural or pharmacological data supporting the three-class model of propofol binding to GABAA receptors.
Three classes of propofol binding sites on GABAA receptors.
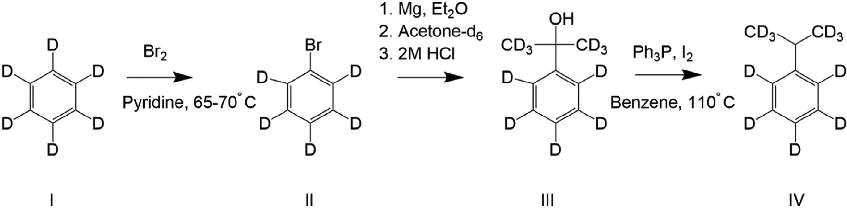
Synthetic scheme for the preparation of cumene-d11 from benzene-d6, used as a deuterated propofol analog for binding site characterization studies on GABAA receptors.
Three classes of propofol binding sites on GABAA receptors.
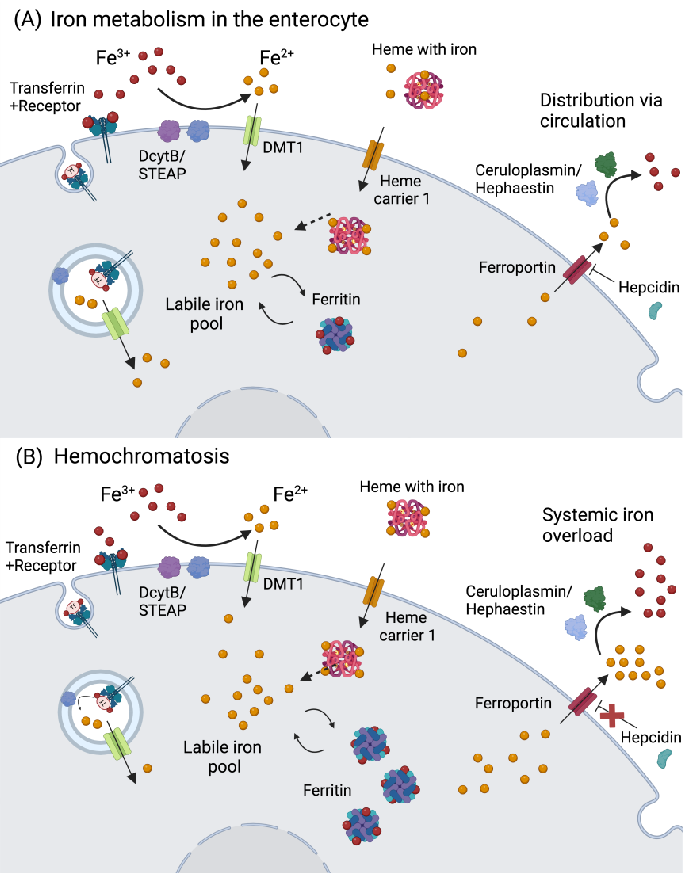
Figure 1. Iron metabolism in the enterocyte and hemochromatosis. (A) Iron is absorbed as Fe2+ through reduction by DcytB/STEAP, and transported via DMT1. In the case iron is bound to …
Metabolic Derangement of Essential Transition Metals and Potential Antioxidant Therapies.
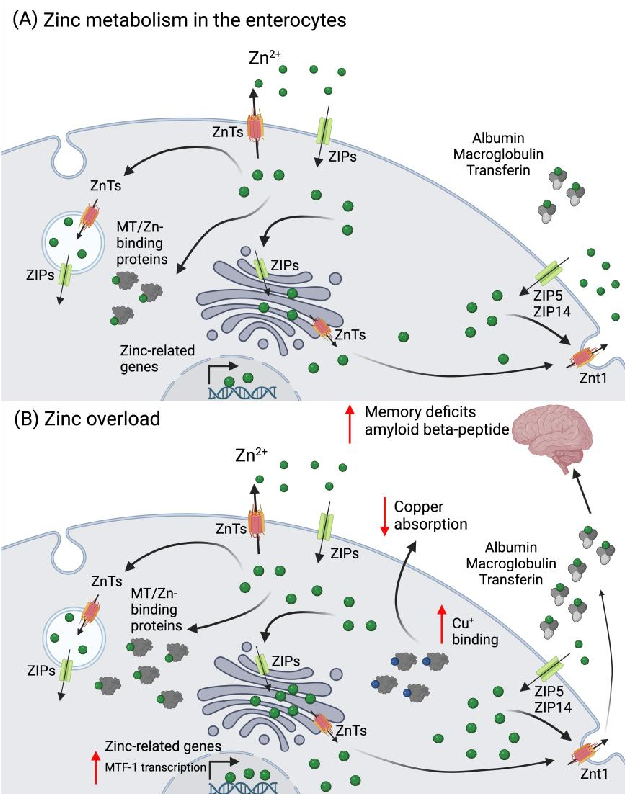
Figure 2. Zinc metabolism in the enterocyte and its overload. (A) Zinc is absorbed as Zn2+ via the ZIPs (mainly ZIP4) and can be excreted into the intestinal lumen by …
Metabolic Derangement of Essential Transition Metals and Potential Antioxidant Therapies.
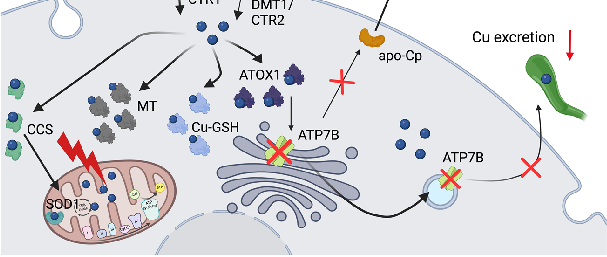
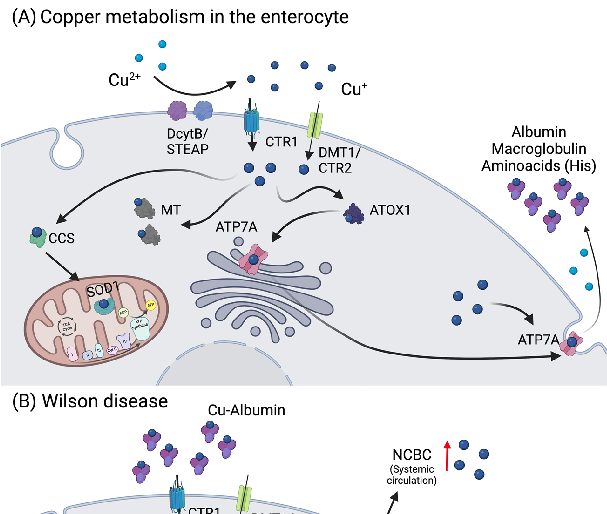
Figure 3. Copper metabolism in the enterocytes and Wilson disease. (A) Copper is reduced to Cu+ by STEAP or DcytB, and transported mainly via CTR1, but also by DMT1/CTR2. Thereupon,
Metabolic Derangement of Essential Transition Metals and Potential Antioxidant Therapies.
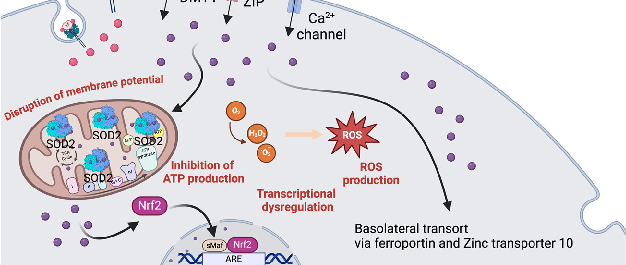
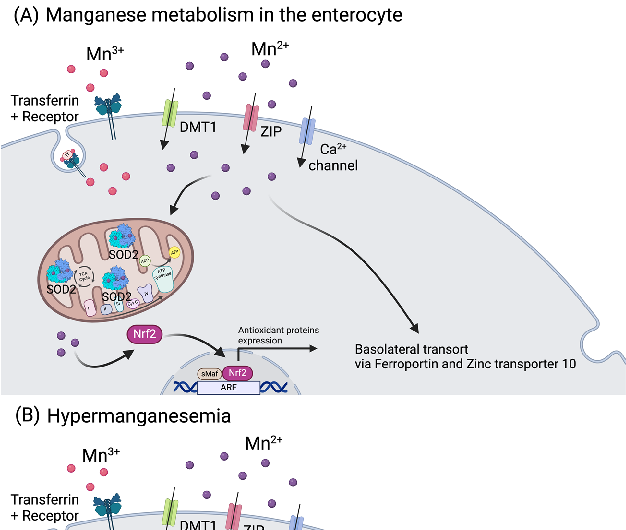
Figure 4. Manganese metabolism and hypermanganesemia. (A) Manganese can be taken up as Mn2+ via DMT1, ZIP, or Ca2+ channels, or as Mn3+ by binding to transferrin. In mitochondria, Mn2+ …
Metabolic Derangement of Essential Transition Metals and Potential Antioxidant Therapies.
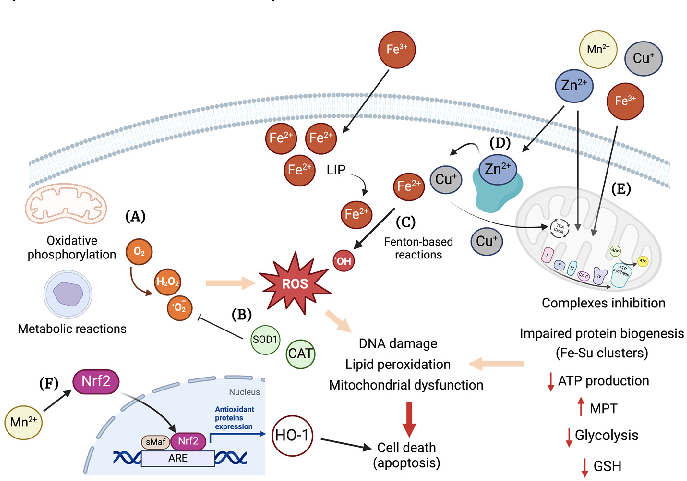
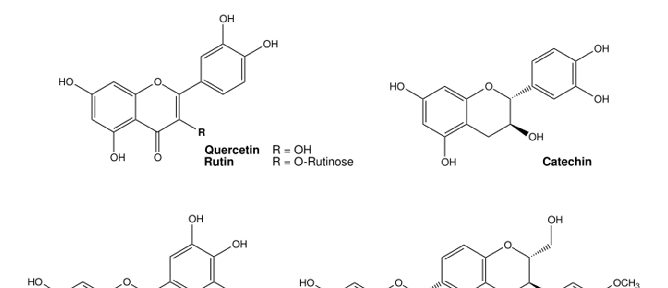
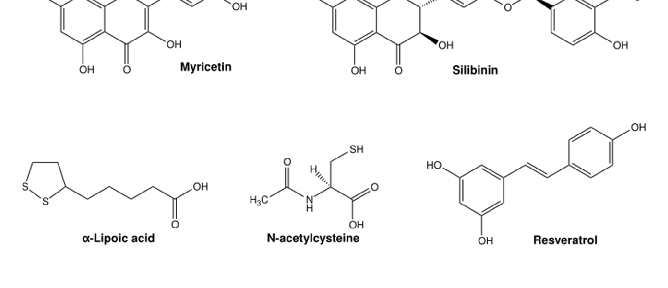
Figure 5. Overview of cellular mechanisms related to toxicity by essential transition metals. (A) Different metabolic pathways in the cell use O2, e.g., oxidative phosphorylation in the mitochondria. The generation …
Metabolic Derangement of Essential Transition Metals and Potential Antioxidant Therapies.