Curcumin Рисунки
87 иллюстрации из рецензируемых исследований
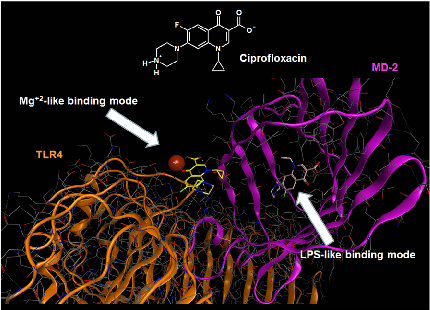
Molecular docking analysis reveals two alternative binding conformations of ciprofloxacin within the TLR4-MD-2 complex binding pocket, suggesting direct physical interaction with the innate immune receptor.
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.
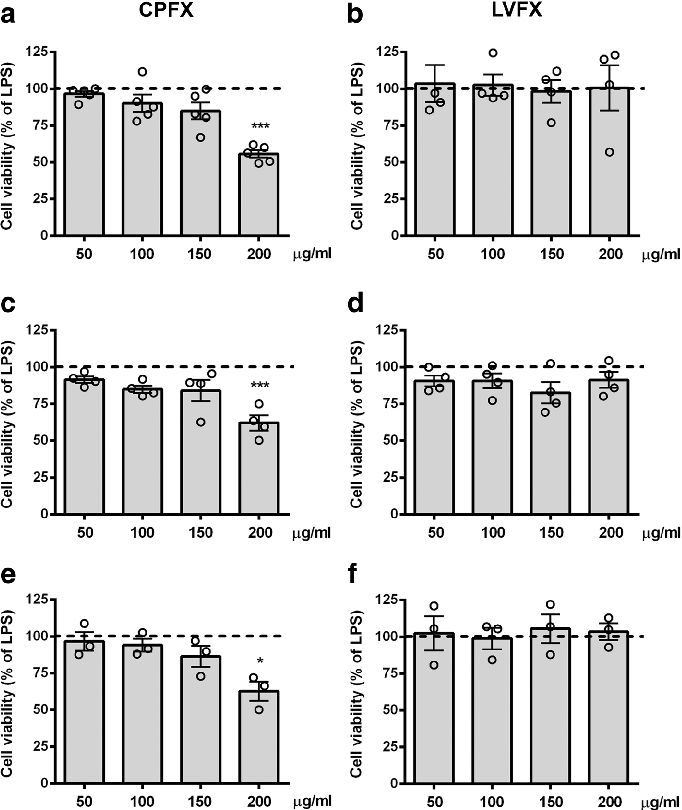
Cell viability assays demonstrate that ciprofloxacin and levofloxacin at the tested concentrations do not significantly reduce microglial survival, confirming that anti-inflammatory effects are not due to cytotoxicity.
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.
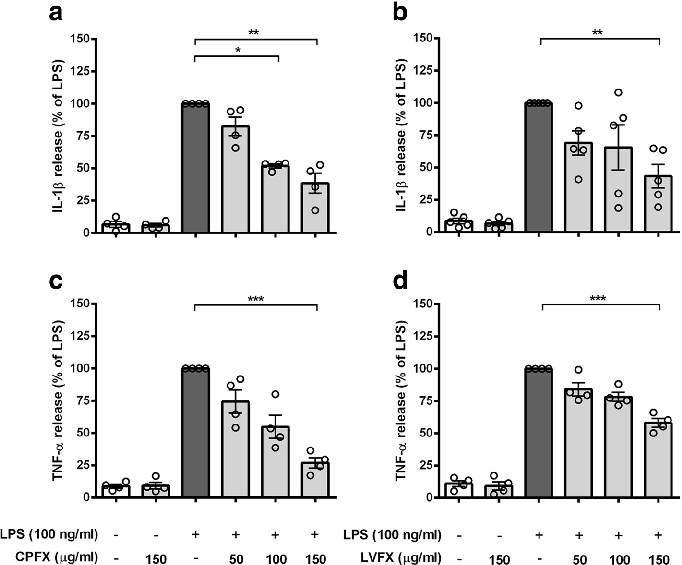
Cytokine release profiles from LPS-stimulated cortical microglia reveal dose-dependent reductions in TNF-alpha and IL-6 following fluoroquinolone treatment.
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.
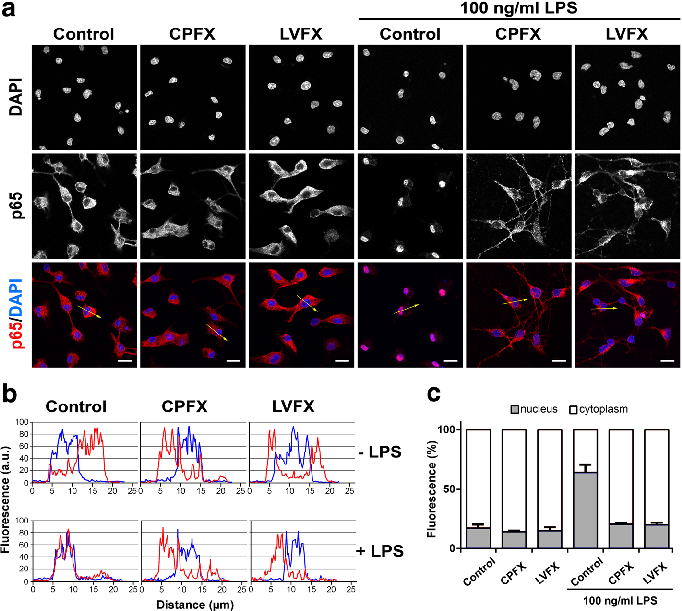
NF-kB nuclear translocation in LPS-stimulated microglia is attenuated by both ciprofloxacin and levofloxacin, as shown by immunofluorescence or reporter gene assays.
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.
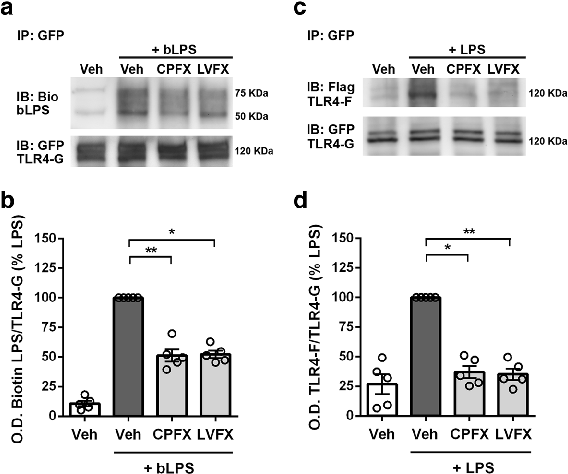
LPS binding and TLR4 dimerization assays in Ba/F3 cells demonstrate that fluoroquinolones interfere with the initial receptor activation step of innate immune signaling.
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.
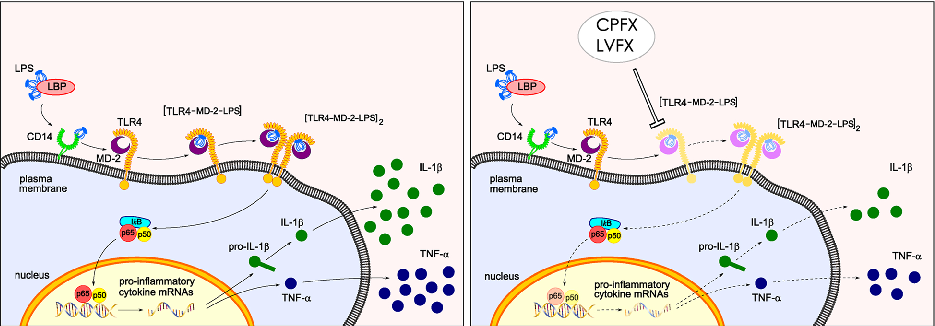
Proposed mechanistic model depicts how ciprofloxacin and levofloxacin target the TLR4-MD-2 complex to block LPS-induced downstream signaling cascades and cytokine production.
Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway.
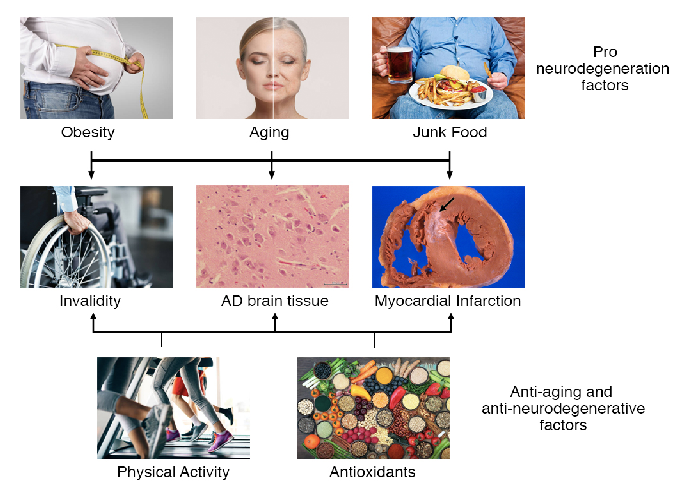
Pro-degeneration factors including aging, obesity, and unhealthy diets can be counterbalanced by physical activity, caloric restriction, and antioxidants. This diagram illustrates how these opposing influences modulate the onset, severity, and duration of neurodegenerative diseases including Alzheimer's, Parkinson's, and ALS.
Dietary habits, lifestyle factors and neurodegenerative diseases.
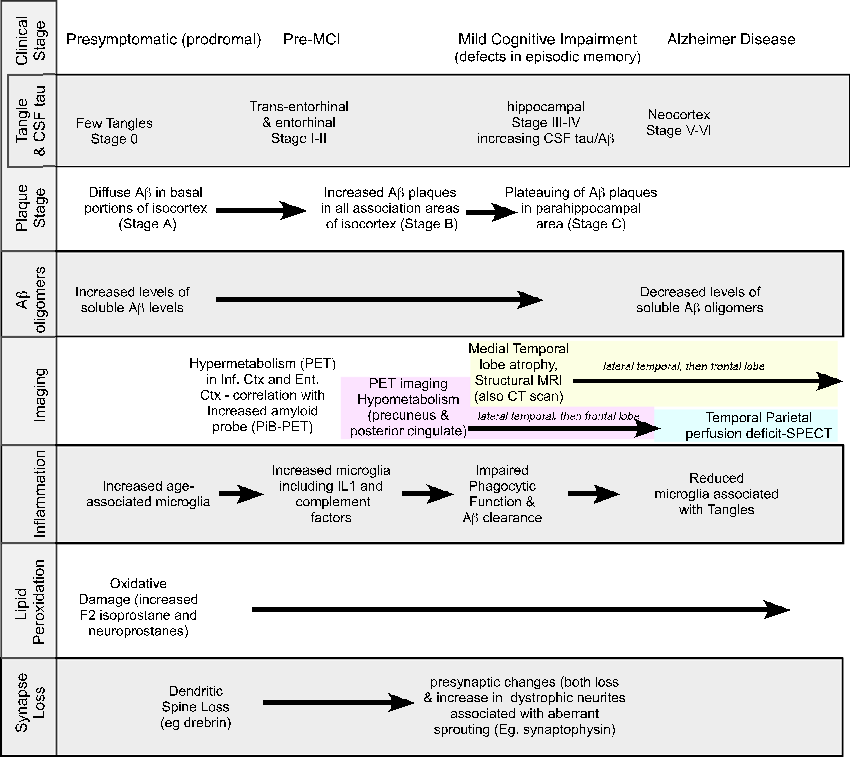
Alzheimer's disease involves a complex pathological cascade initially triggered by amyloid-beta accumulation or aberrant APP processing. This figure argues for pleiotropic interventions that simultaneously target multiple pathological mechanisms rather than single molecular targets.
Why pleiotropic interventions are needed for Alzheimer's disease.
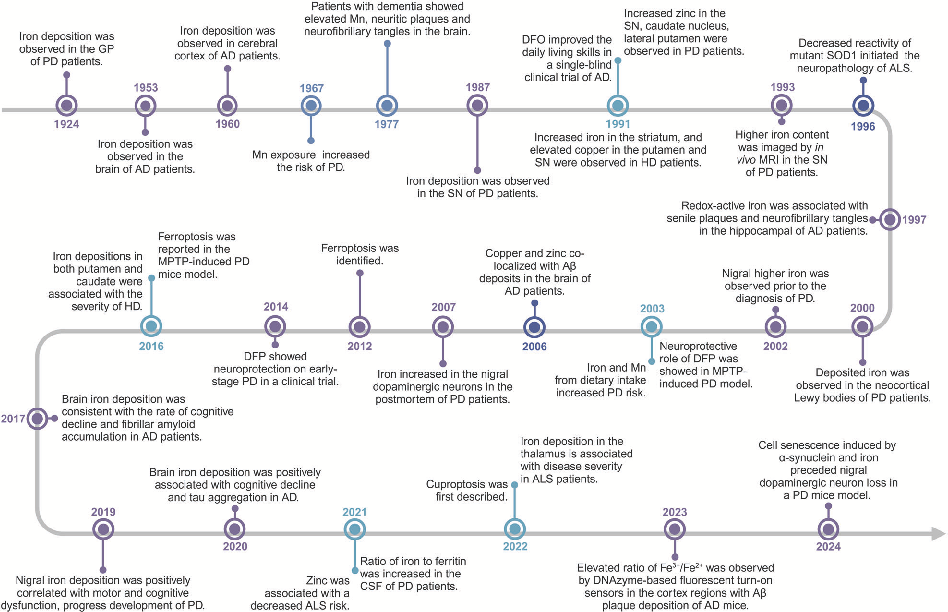
Molecular pathways of iron metabolism and ferroptosis in the context of neurodegenerative disease. Excessive iron accumulation in neurons can trigger lipid peroxidation and cell death through ferroptotic mechanisms.
Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases.
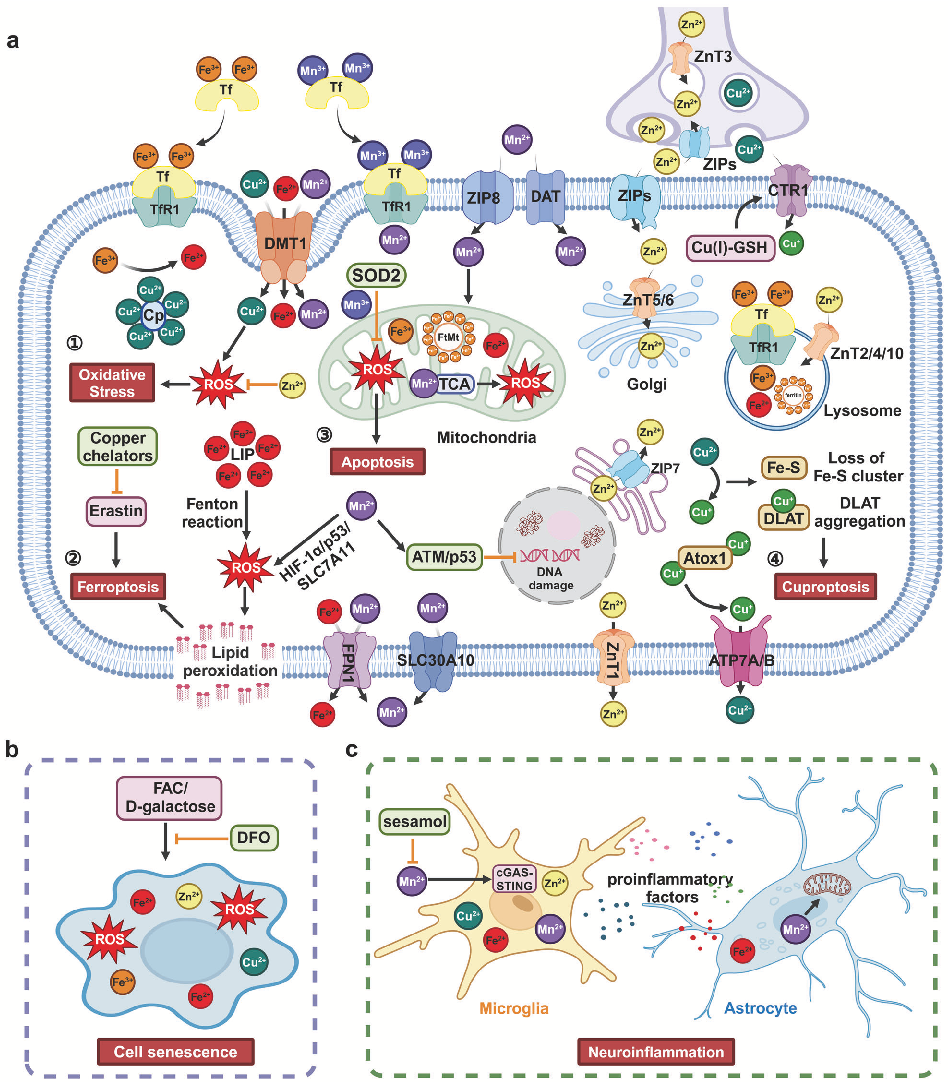
Schematic of copper homeostasis and cuproptosis mechanisms relevant to neurodegeneration. Copper imbalance has been implicated in the pathogenesis of Wilson's disease and may contribute to Alzheimer's disease progression.
Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases.
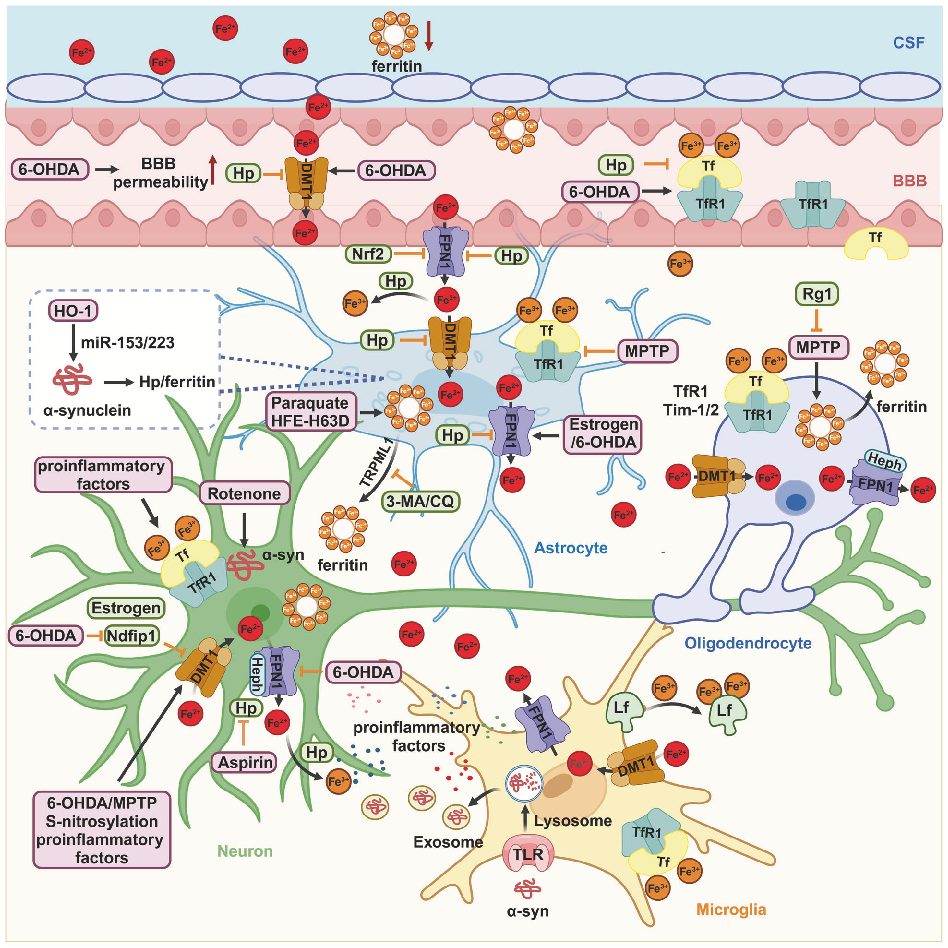
Illustration of zinc and manganese transport and regulatory mechanisms in the central nervous system, highlighting how disruptions in metal ion balance may accelerate neurodegenerative processes.
Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases.
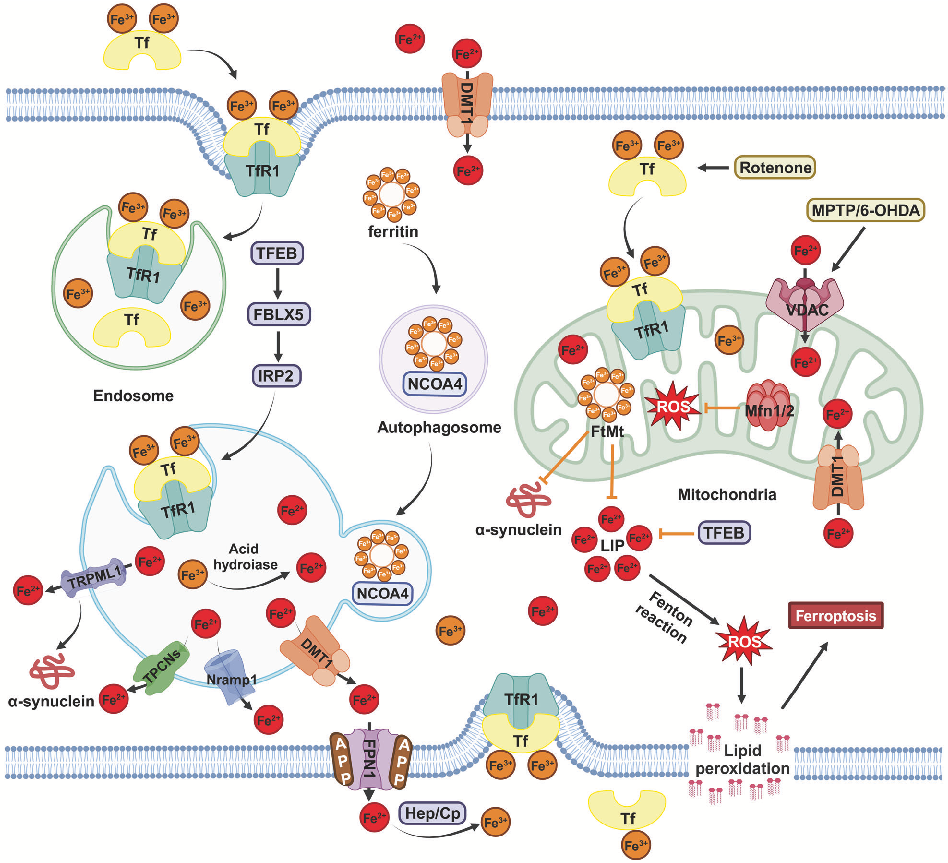
Summary of therapeutic strategies targeting metal ion dysregulation in neurodegenerative diseases, including iron chelation, antioxidant supplementation, and metal transporter modulation.
Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases.
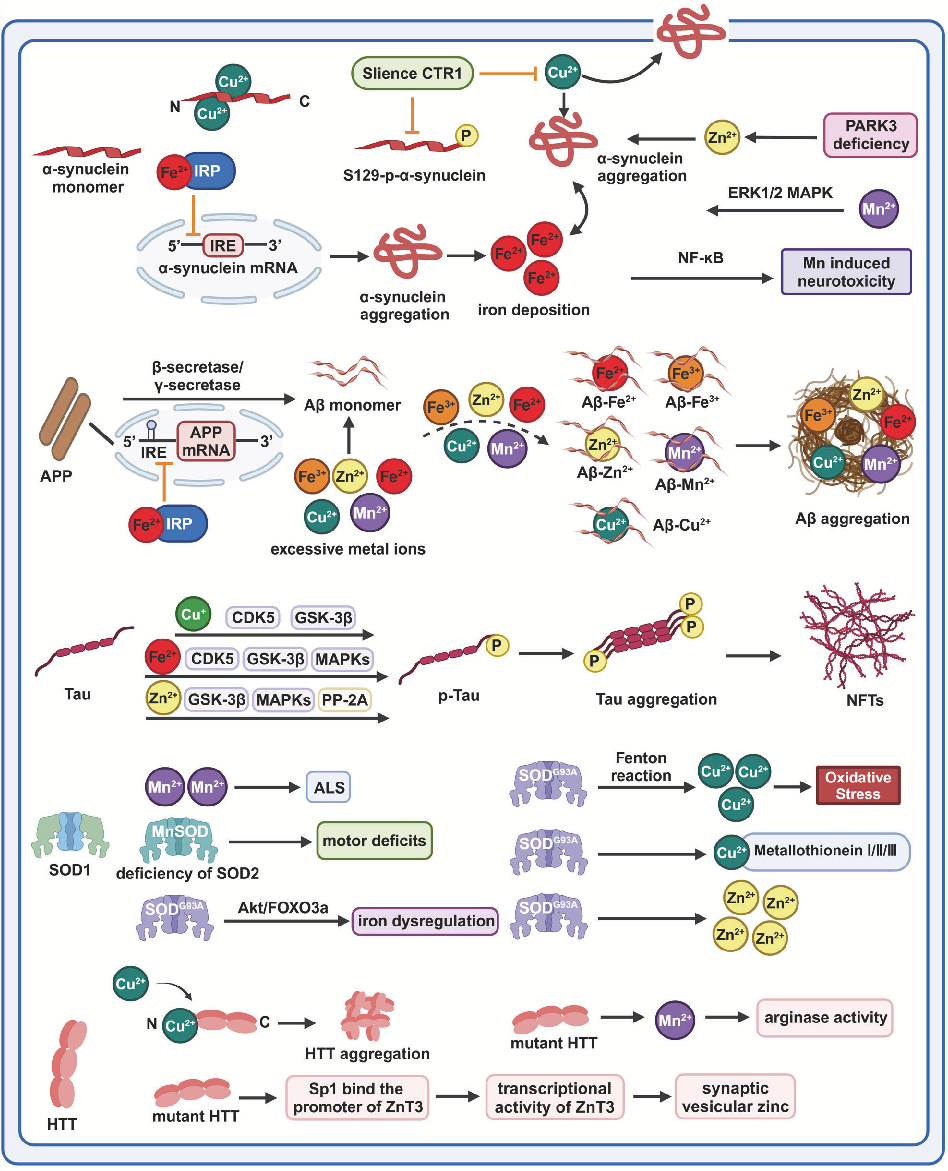
Integrative model linking metal ion imbalance to multiple neurodegenerative disease mechanisms including oxidative stress, ferroptosis, cuproptosis, cellular senescence, and neuroinflammation.
Homeostasis and metabolism of iron and other metal ions in neurodegenerative diseases.
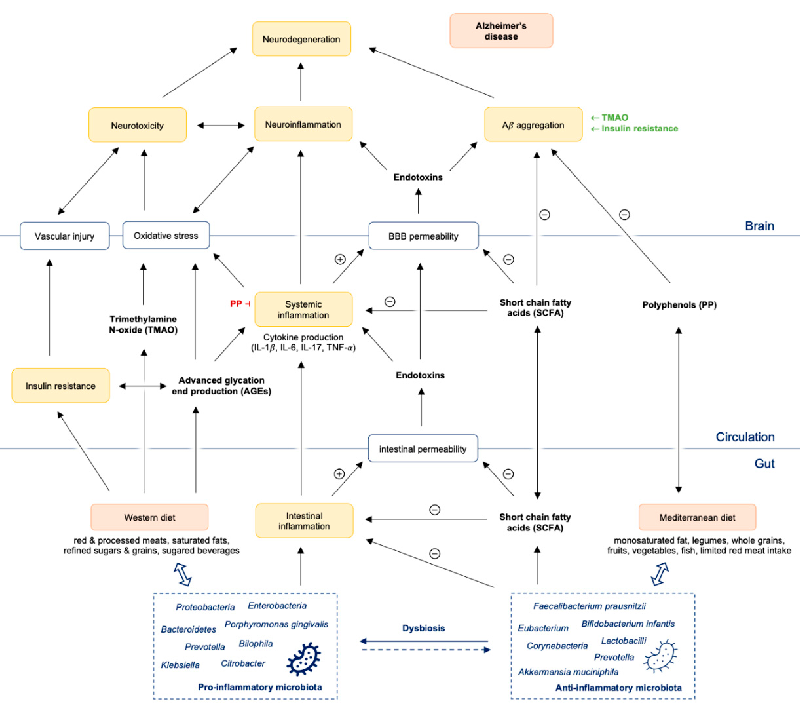
A pathway diagram illustrates how gut dysbiosis promotes intestinal and systemic inflammation, leading to amyloid-beta aggregation, neuroinflammation, and ultimately neurodegeneration in Alzheimer's disease. The cascade connects microbial imbalance to blood-brain barrier compromise and central nervous system pathology.
The Immunopathogenesis of Alzheimer's Disease Is Related to the Composition of Gut …
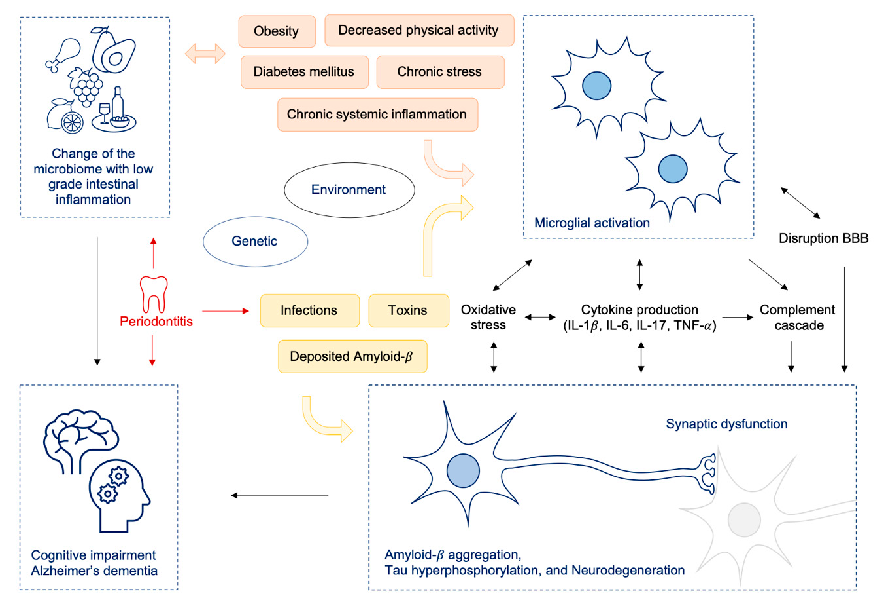
Western diet-induced microbiome changes are mapped to intestinal dysbiosis, low-grade gut inflammation, and increased permeability of both the intestinal barrier and blood-brain barrier. The resulting systemic inflammatory state is linked to neuroinflammatory processes implicated in Alzheimer's disease progression.
The Immunopathogenesis of Alzheimer's Disease Is Related to the Composition of Gut …
Major pharmacotherapeutic targets in Alzheimer's disease are mapped, including amyloid-beta aggregation, tau hyperphosphorylation, neuroinflammation, and cholinergic dysfunction as key intervention points.
Review of Pharmacotherapeutic Targets in Alzheimer's Disease and Its Management Using Traditional …
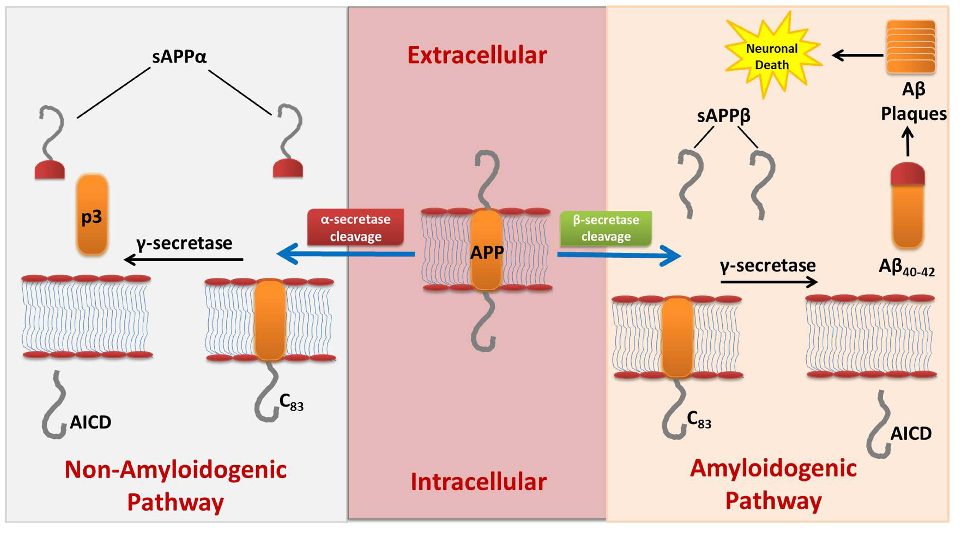
The amyloid cascade hypothesis and its therapeutic targets are illustrated, showing how beta-secretase and gamma-secretase inhibitors, along with anti-amyloid antibodies, aim to reduce pathological plaque formation.
Review of Pharmacotherapeutic Targets in Alzheimer's Disease and Its Management Using Traditional …
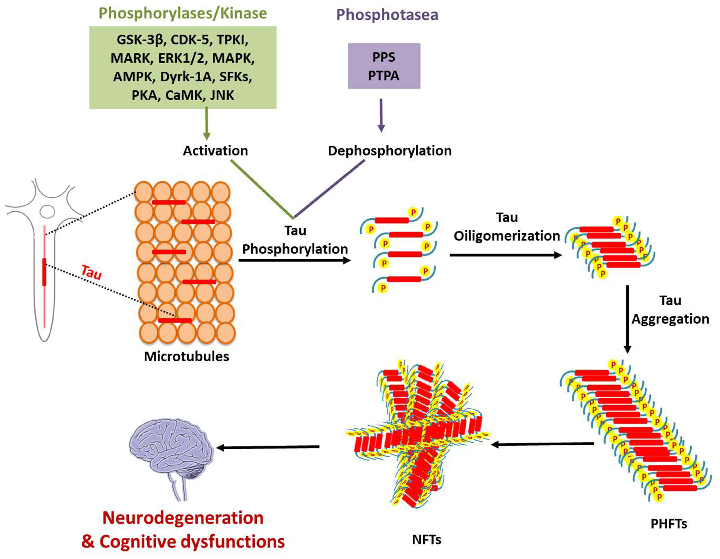
Tau protein pathology and potential therapeutic interventions are depicted, including kinase inhibitors and immunotherapy approaches targeting neurofibrillary tangle formation in Alzheimer's disease.
Review of Pharmacotherapeutic Targets in Alzheimer's Disease and Its Management Using Traditional …
Neuroinflammatory pathways in Alzheimer's disease and anti-inflammatory therapeutic targets are outlined, showing microglial activation, cytokine cascades, and potential points of pharmacological intervention.
Review of Pharmacotherapeutic Targets in Alzheimer's Disease and Its Management Using Traditional …
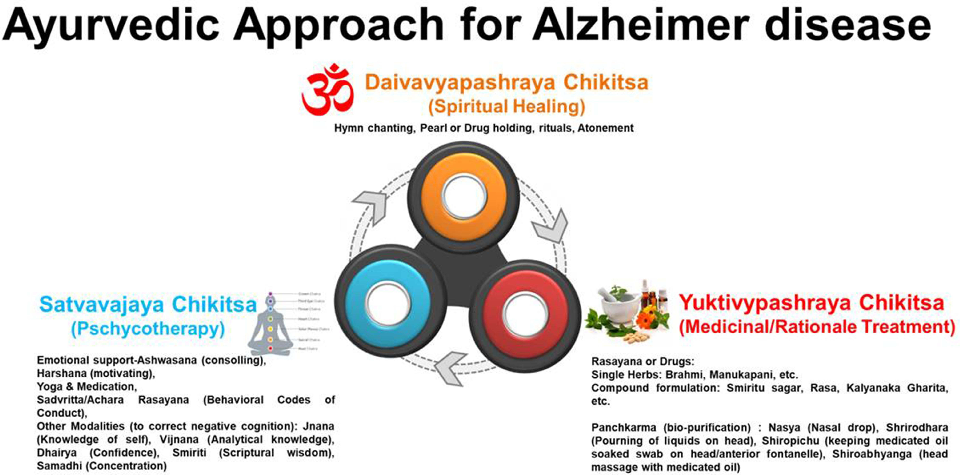
Traditional medicinal plants investigated for Alzheimer's management are catalogued with their bioactive compounds and proposed neuroprotective mechanisms, including antioxidant and anti-inflammatory activities.
Review of Pharmacotherapeutic Targets in Alzheimer's Disease and Its Management Using Traditional …
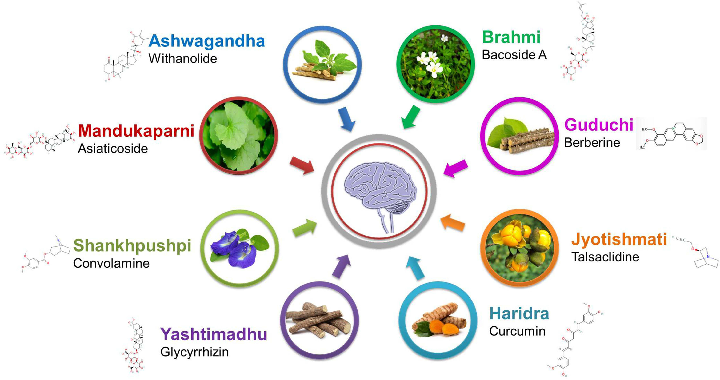
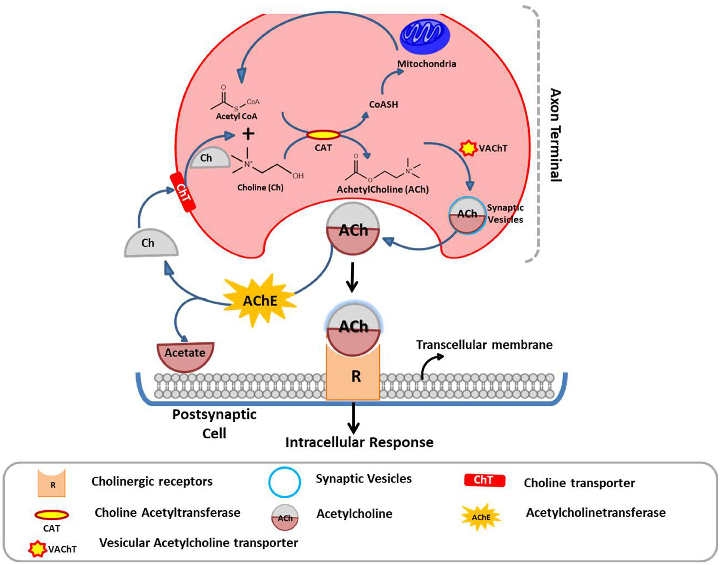
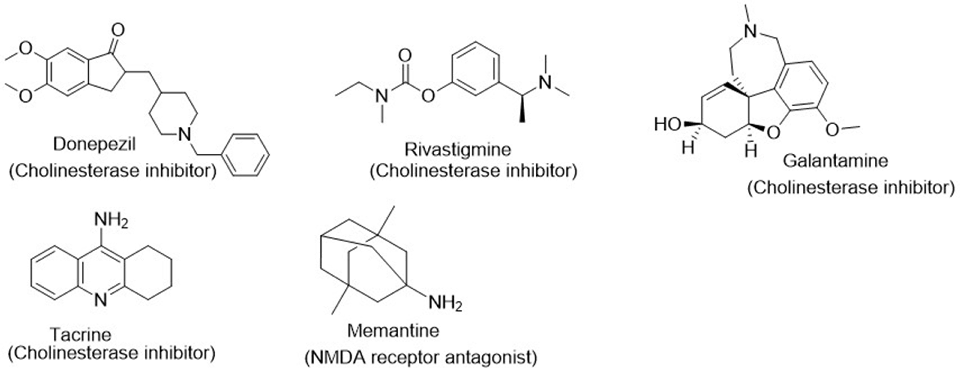
Cholinergic system dysfunction in Alzheimer's disease and current cholinesterase inhibitor therapies are illustrated, showing how acetylcholine deficiency contributes to cognitive decline.
Review of Pharmacotherapeutic Targets in Alzheimer's Disease and Its Management Using Traditional …
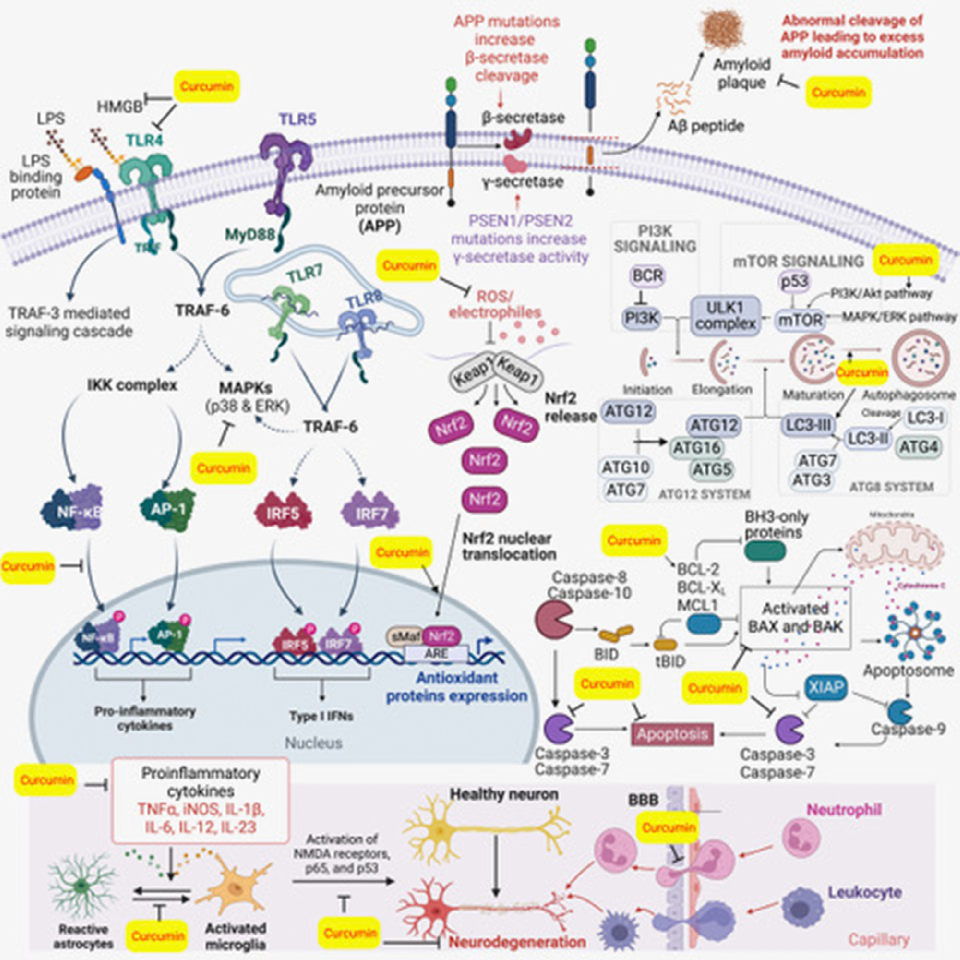
Major inflammatory pathways associated with neurological disorders and curcumin's potential to restore deregulated signaling are depicted. NF-kB, Nrf2, and inflammasome pathways are central to curcumin's neuroprotective mechanism.
Curcumin, inflammation, and neurological disorders: How are they linked?
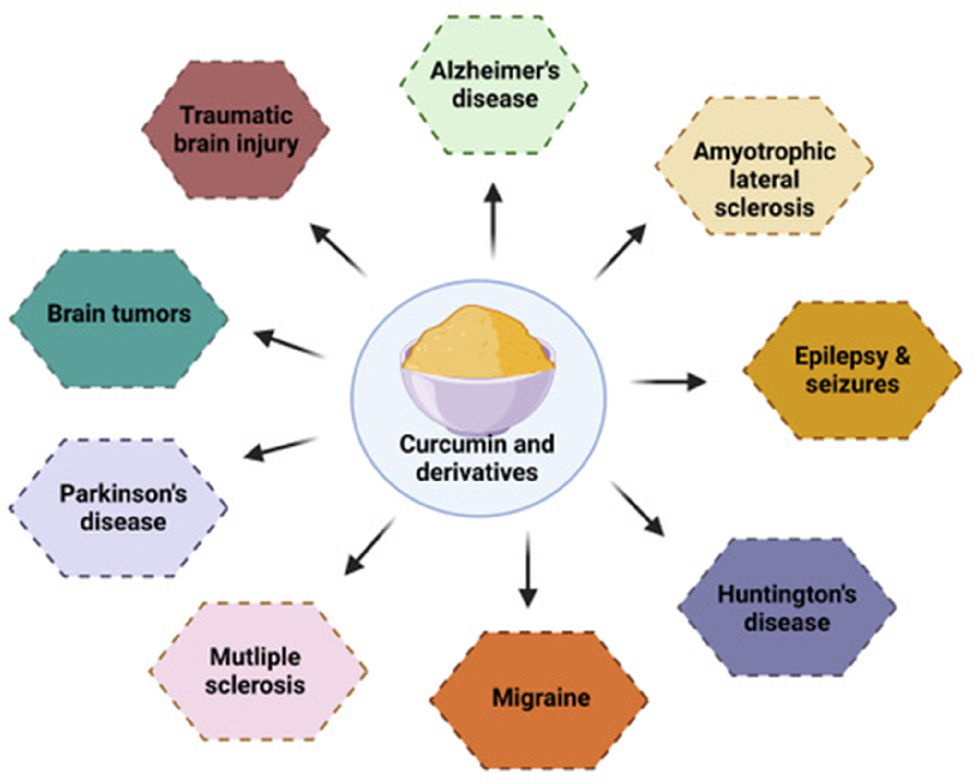
Curcumin activity against various neurodegenerative diseases is illustrated, spanning Alzheimer's disease, Parkinson's disease, Huntington's disease, and amyotrophic lateral sclerosis. Distinct molecular targets are engaged in each condition.
Curcumin, inflammation, and neurological disorders: How are they linked?
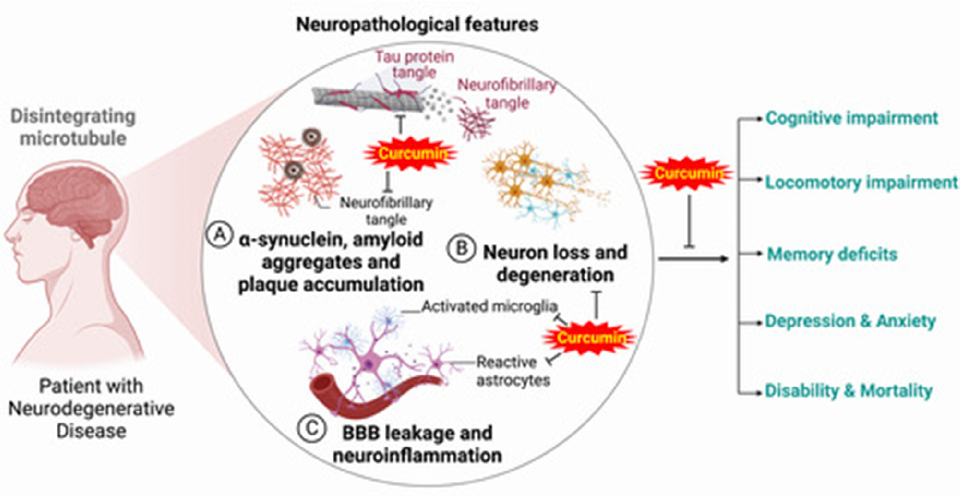
Neuropathological features associated with commonly occurring neurodegenerative diseases and their clinical outcomes are depicted. Protein aggregation, neuroinflammation, and oxidative stress represent shared pathological hallmarks.
Curcumin, inflammation, and neurological disorders: How are they linked?
Страница 1 из 4